13.1: Prefix
- Page ID
- 2410
In the last five chapters, we have seen a variety of reaction mechanisms - nucleophilic substitutions, phosphoryl transfers, carbonyl additions, and acyl substitutions - in which bonds were broken and new bonds were formed. In each case, these events involved bonds between carbon (or phosphorus) and a heteroatom - usually oxygen or nitrogen, but occasionally sulfur or a halide. We have yet to see a reaction where a carbon-carbon bond is formed or broken. To put it another way, although we have seen many examples of carbon electrophiles, we have not seen a reaction with a carbon atom acting as a nucleophile or a leaving group.
In order for an atom to be nucleophilic, it must have a pair of electrons available for bonding (the exception is reactions involving free radicals, which we will consider in chapter 17). Heteroatom nucleophiles - the oxygen atoms of alcohols, for example - have lone pairs of electrons. In a carbon nucleophile, the reactive electrons might be held in a relatively 'loose' bond, like the pi bond of an alkene.
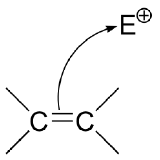
In chapter 15, we will study reactions in which the pi electrons of alkenes act in a nucleophilic fashion.
The other possible way that a carbon could be nucleophilic is if it has a lone pair of non-bonding electrons - in other words, if it is a carbanion.

Likewise, you would expect that a carbon leaving group would be a carbanion, because it takes two electrons with it when it departs. But aren't carbanions highly unstable? Aren't they terrible leaving groups? Aren't they far too basic to be relevant intermediate species in the aqueous, pH neutral environment of a living cell? They certainly are, if the negative charge is 'locked', or localized, on a single carbon, which is an extremely unstable situation. In order for a carbanion to be a reasonable participant in an aqueous reaction - a nucleophile or leaving group - there clearly must be some kind of powerful stabilizing factor involved which acts to delocalize the unbonded electron pair, and its associated negative charge, away from the carbon atom.
That stabilizing factor, as you may already have predicted, is more often than not an adjacent carbonyl group. Recall from section 7.5 that protons at the alpha position - on carbons directly adjacent to a carbonyl - are somewhat acidic, because the negative charge on the conjugate base is stabilized by resonance with the carbonyl oxygen.
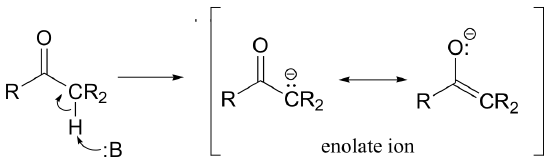
This is the so-called 'enolate' form of stabilized carbanion that, in biochemical reactions, most often serves as either a nucleophile or leaving group. However, there are other ways in which carbanion reaction intermediates can be stabilized. In the next two chapters, we will explore a variety of reactions that proceed through resonance-stabilized carbanion intermediates. In this chapter, the focus will be on two very important carbon-carbon bond-forming / bond breaking events known as 'aldol' and 'Claisen' reactions, in which stabilized carbanions act as nucleophiles or leaving groups. First, however, we will be introduced to the concept of tautomerization, and then see some examples of biochemical isomerization reactions in which two constitutional isomers or stereoisomers are interconverted via stabilized carbanion intermediates.


