6.6: Cómo funcionan los enzimas
- Page ID
- 2357
So far, what we have learned about protein structure applies to all kinds of proteins with all kinds of functions. It is time now to focus on enzymes, those proteins whose role it is to catalyze the myriad organic (and inorganic) reactions that occur in living things. We already know, in very general terms, what catalysis is: it is the lowering of the activation energy of a reaction that results in rate acceleration. Using slightly different terms to describe the same thing, we can also speak of catalysis as ‘transition state stabilization’. Now, we need to think about just how an enzyme lowers a reaction’s activation energy. We will introduce here some of the most fundamental concepts of enzyme catalysis, and we will continue to encounter specific examples throughout the rest of the text as we learn about different organic reaction mechanisms.
6.5A: The active site
A critical element in the three-dimensional structure of any enzyme is the presence of an ‘active site’, which is a pocket, usually located in the interior of the protein, that serves as a docking point for the enzyme’s substrate(s) (‘substrate’ is the term that biochemists use for a reactant molecule in an enzyme-catalyzed reaction). It is inside the active site pocket that enzymatic catalysis occurs. Shown below is an image of the glycolytic enzyme fructose-1,6-bisphosphate aldolase, with its substrate bound inside the active site pocket.
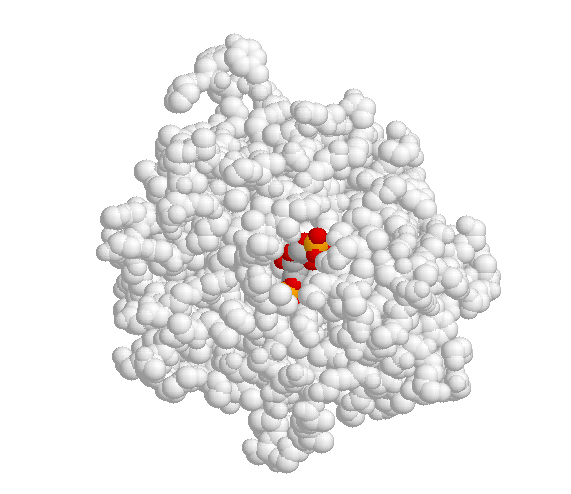
When the substrate binds to the active site, a large number of noncovalent interactions form with the amino acid residues that line the active site. The shape of the active site, and the enzyme-substrate interactions that form as a result of substrate binding, are specific to the substrate-enzyme pair: the active site has evolved to 'fit' one particular substrate and to catalyze one particular reaction. Other molecules do not fit in this active site nearly so well as fructose 1,6-bisphosphate.
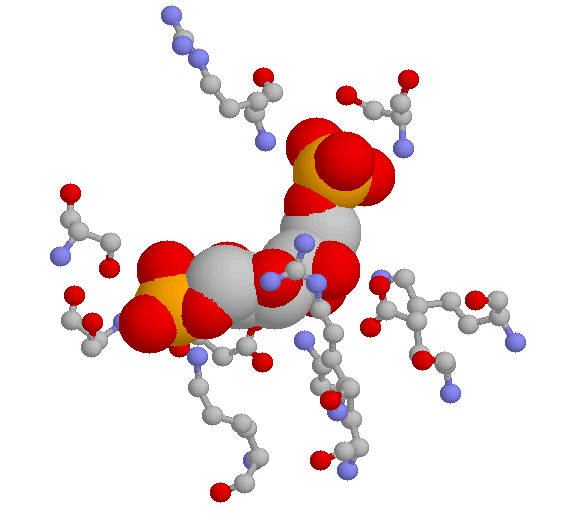
Here are two close-up views of the same active site pocket, showing some of the specific hydrogen-bonding interactions between the substrate and active site amino acids. The first image below is a three-dimensional rendering directly from the crystal structure data. The substrate is shown in 'space-filling' style, while the active site amino acids are shown in the 'ball and stick' style. Hydrogens are not shown. The color scheme is grey for carbon, red for oxygen, blue for nitrogen, and orange for phosphorus.
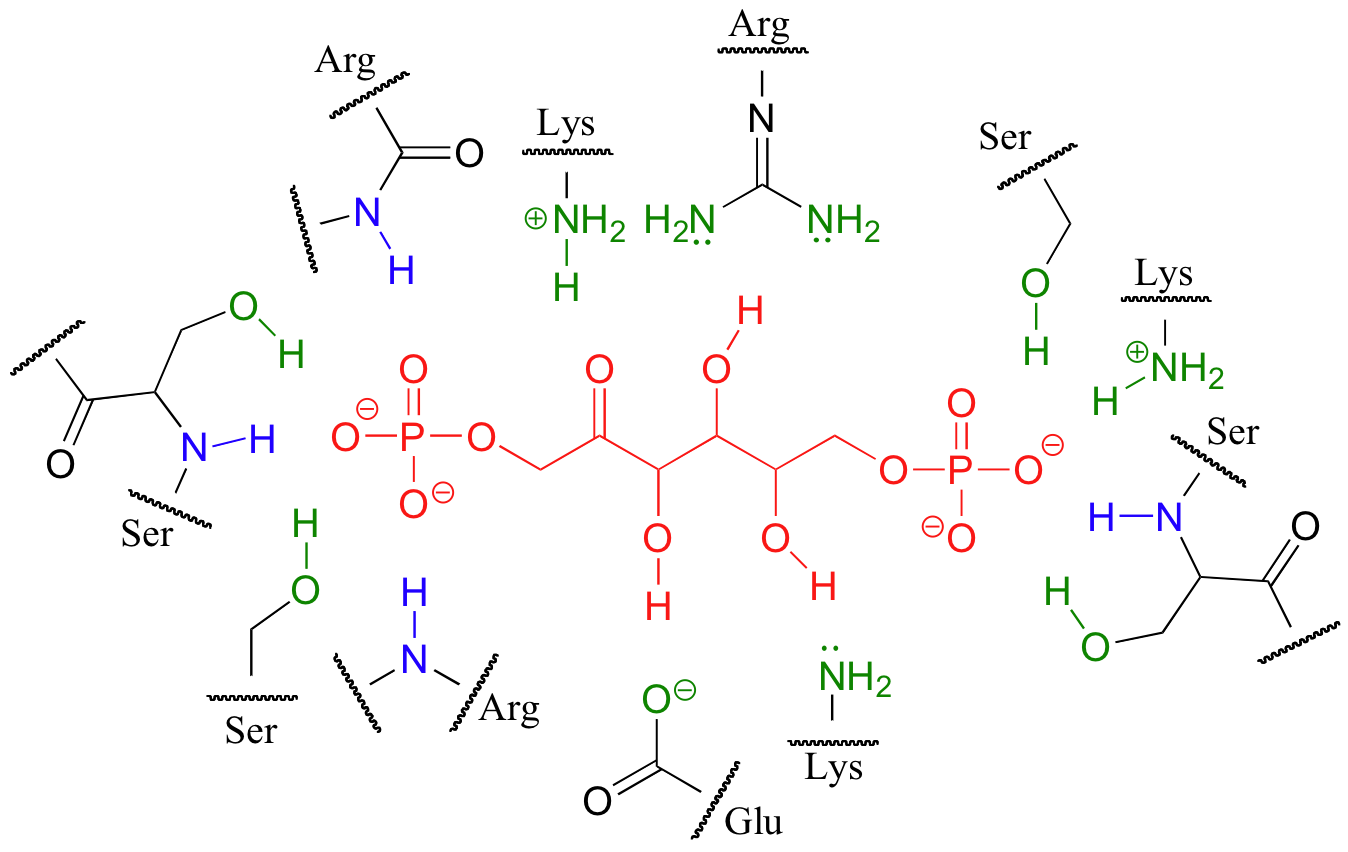
Below is a two-dimensional picture of the substrate (colored red) surrounded by hydrogen-bonding active site amino acids. Notice that both main chain and side chain groups contribute to hydrogen bonding: in this figure, main chain H-bonding groups are colored blue, and side chain H-bonding groups are colored green.
Looking at the last three images should give you some appreciation for the specific manner in which a substrate fits inside its active site.
6.5B: Transition state stabilization
One of the most important ways that an enzyme catalyzes any given reaction is through entropy reduction: by bringing order to a disordered situation (remember that entropy is a component of Gibbs Free Energy, and thus a component of the activation energy). Let’s turn again to our previous example (from section 6.1C) of a biochemical nucleophilic substitution reaction, the methylation of adenosine in DNA. The reaction is shown below with non-reactive sections of the molecules depicted by variously shaped 'bubbles' for the sake of simplicity.

In order for this reaction to occur, the two substrates (reactants) must come into contact in precisely the right way. If they are both floating around free in solution, the likelihood of this occurring is very small – the entropy of the system is simply too high. In other words, this reaction takes place very slowly without the help of a catalyst.
Here’s where the enzyme’s active site pocket comes into play. It is lined with various functional groups from the amino acid main and side chains, and has a very specific three-dimensional architecture that has evolved to bind to both of the substrates. If the SAM molecule, for example, diffuses into the active site, it can replace its (favorable) interactions with the surrounding water molecules with (even more favorable) new interactions with the functional groups lining the active site.
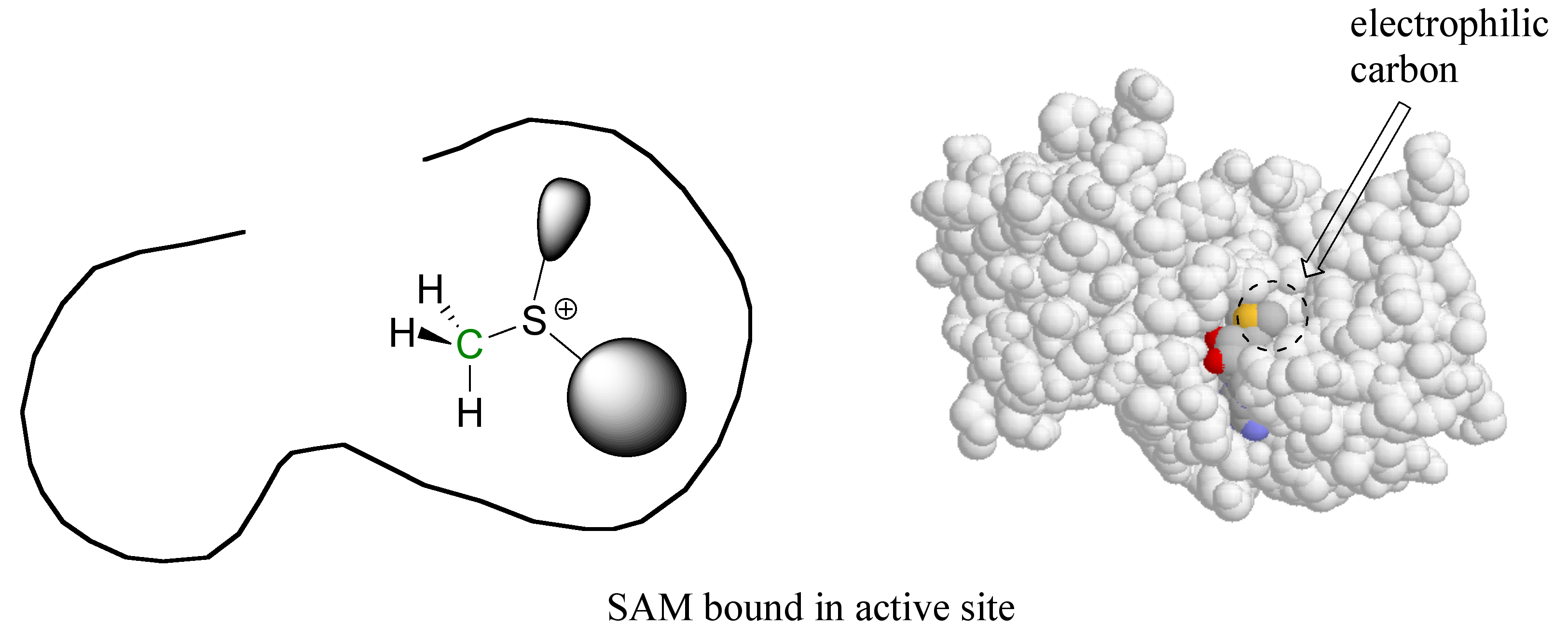
In a sense, SAM is moving from one solvent (water) to another 'solvent' (the active site), where many new energetically favorable interactions are possible. Remember: these new contacts between SAM and the active site groups are highly specific to SAM and SAM alone – no other molecule can ‘fit’ so well in this precise active site environment, and thus no other molecule will be likely to give up its contacts to water and bind to the active site.
The second substrate also has a specific spot reserved in the active site. (Because in this case the second substrate is a small segment of a long DNA molecule, the DNA-binding region of the active site is more of a 'groove' than a 'pocket').
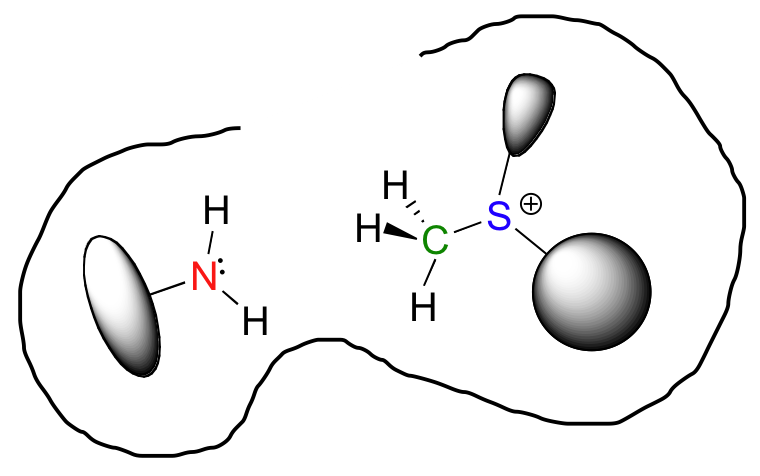
So now we have both substrates bound in the active site. But they are not just bound in any random orientation – they are specifically positioned relative to one another so that the nucleophilic nitrogen is held very close to the electrophilic carbon, with a free path of attack. What used to be a very disordered situation – two reactants diffusing freely in solution – is now a very highly ordered situation, with everything set up for the reaction to proceed. This is what is meant by entropy reduction: the entropic component of the energy barrier has been lowered.
Looking a bit deeper, though, it is not really the noncovalent interaction between enzyme and substrate that are responsible for catalysis. Remember: all catalysts, enzymes included, accelerate reactions by lowering the energy of the transition state. With this in mind, it should make sense that the primary job of an enzyme is to maximize favorable interactions with the transition state, not with the starting substrates. This does not imply that enzyme-substrate interactions are not strong, rather that enzyme-TS interactions are far stronger, often by several orders of magnitude. Think about it this way: if an enzyme were to bind to (and stabilize) its substrate(s) more tightly than it bound to (and stabilized) the transition state, it would actually slow down the reaction, because it would be increasing the energy difference between starting state and transition state. The enzyme has evolved to maximize favorable noncovalent interactions to the transition state: in our example, this is the state in which the nucleophilic nitrogen is already beginning to attack the electrophilic carbon, and the carbon-sulfur bond has already begun to break.
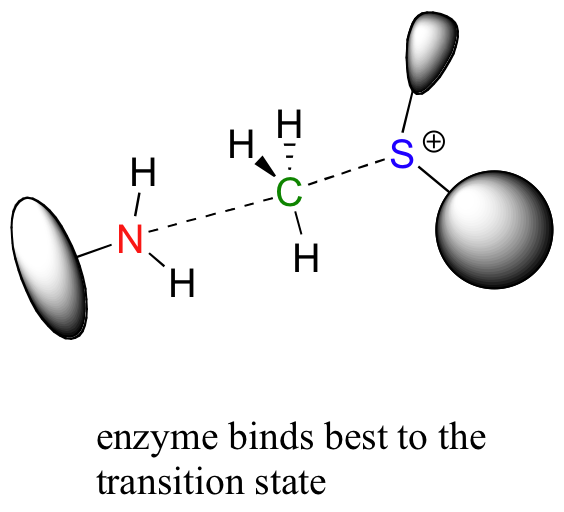
In many enzymatic reactions, certain active site amino acid residues contribute to catalysis by increasing the reactivity of the substrates. Often, the catalytic role is that of acid and/or base. In our DNA methylation example, the nucleophilic nitrogen is deprotonated by a nearby aspartate side chain as it begins its nucleophilic attack on the methyl group of SAM. We will study nucleophilicity in greater detail in chapter 8, but it should make intuitive sense that deprotonating the amine increases the electron density of the nitrogen, making it more nucleophilic. Notice also in the figure below that the main chain carbonyl of an active site proline forms a hydrogen bond with the amine, which also has the effect of increasing the nitrogen's electron density and thus its nucleophilicity (Nucleic Acids Res. 2000, 28, 3950).

How does our picture of enzyme catalysis apply to multi-step reaction mechanisms? Although the two-step nucleophilic substitution reaction between tert-butyl chloride and hydroxide (section 6.1C) is not a biologically relevant process, let’s pretend just for the sake of illustration that there is a hypothetical enzyme that catalyzes this reaction.

The same basic principles apply here: the enzyme binds best to the transition state. But therein lies the problem: there are two transition states! To which TS does the enzyme maximize its contacts?
Recall that the first step – the loss of the chloride leaving group to form the carbocation intermediate – is the slower, rate-limiting step. It is this step that our hypothetical enzyme needs to accelerate if it wants to accelerate the overall reaction, and it is thus the energy of TS1 that needs to be lowered.
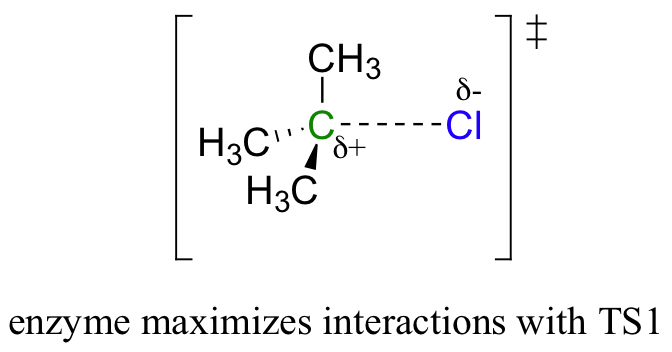
By Hammond’s postulate, we also know that the intermediate I is a close approximation of TS1. So the enzyme, by stabilizing the intermediate, will also stabilize TS1 (as well as TS2) and thereby accelerate the reaction.
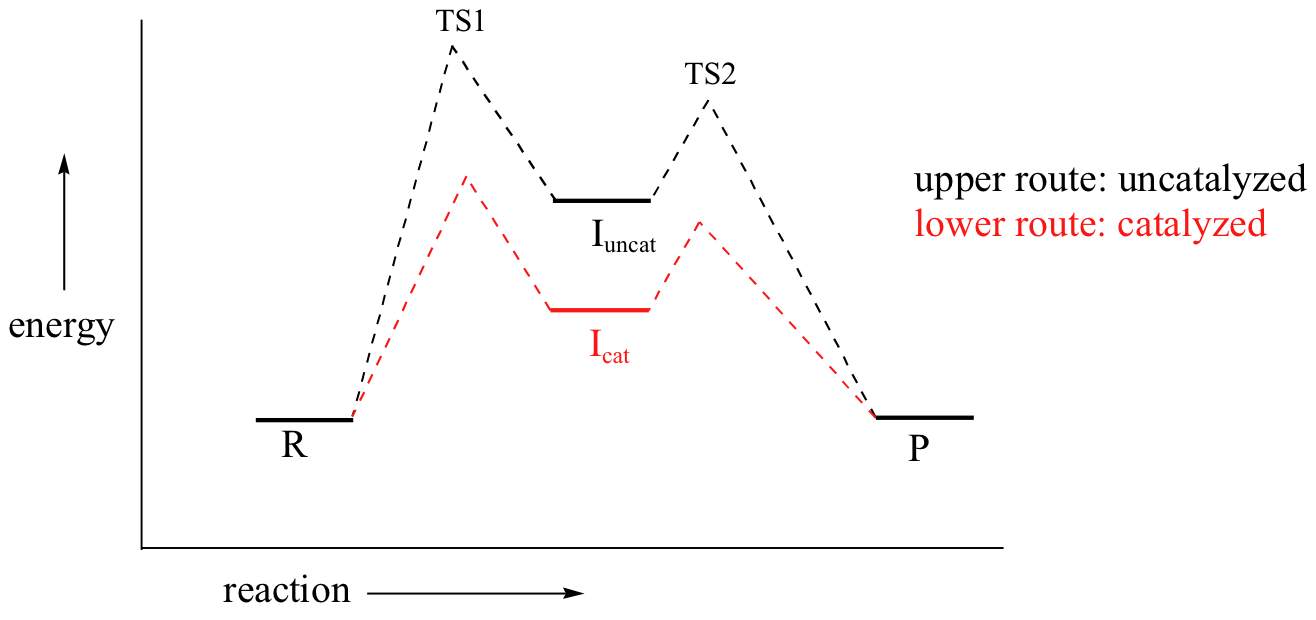
If you read scientific papers about enzyme mechanisms, you will often see researchers discussing how an enzyme stabilizes a reaction intermediate. By virtue of Hammond's postulate, they are, at the same time, talking about how the enzyme lowers the energy of the transition state.
An additional note: although we have in this section been referring to SAM as a 'substrate' of the DNA methylation reaction, it is also often referred to as a coenzyme, or cofactor. These terms are used to describe small (relative to protein and DNA) biological organic molecules that bind specifically in the active site of an enzyme and help the enzyme to do its job. In the case of SAM, the job is methyl group donation. In addition to SAM, we will see many other examples of coenzymes in the coming chapters, a number of which - like ATP (adenosine triphosphate), coenzyme A, thiamine, and flavin - you have probably heard of before. The full structures of some common coenzymes are shown in table 6 in the tables section.
6.5C: Site-directed mutagenesis
Biochemists frequently use a powerful technique called site-directed mutagenesis in order to learn more about the role played by a particular amino acid residue or group of residues in a protein of interest. The technical details of site-directed mutagenesis are beyond the scope of this text, but the end result is not hard to understand: essentially, a researcher is able to change any amino acid in a protein to any other amino acid. If we are interested in the role played by the phenol group in an active site tyrosine, for example, we might use site-directed mutagenesis to produce a mutant protein in which this tyrosine has been changed to a phenylalanine, thus preserving the phenyl group but specifically deleting the phenol.
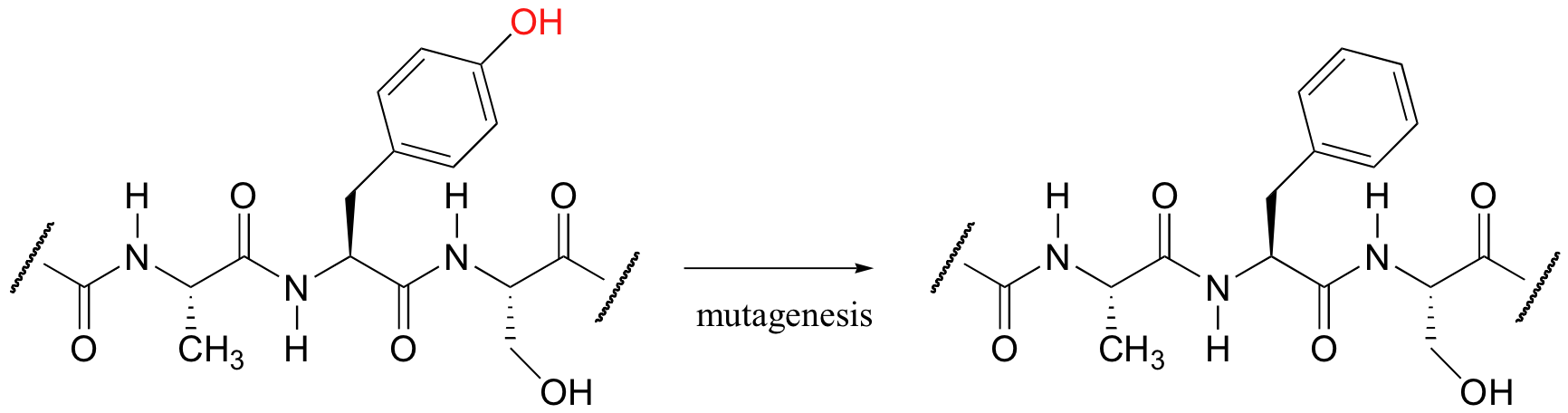
If the mutant protein behaves differently from the 'wild type’ (natural) protein - if it loses all activity, for example - then we can infer that the phenol group plays some important role. In some cases, researchers have even been able to change the product or substrate specificity of an enzymatic reaction using site-directed mutagenesis. The technique has become an indispensable tool for researchers studying enzyme-catalyzed organic reactions.
6.5D: Enzyme inhibitors
The action of enzymes can be slowed or shut down completely by agents called inhibitors. Although there are several different modes of enzyme inhibition, we will focus here on two of the most important: competitive inhibition and irreversible inhibition. In competitive inhibition, a molecule with a structure that closely resembles that of the substrate, intermediate, or product of a particular enzymatic reaction is able to enter and bind (through noncovalent interactions) to the enzyme's active site. This 'imposter' molecule, as long as it stays in the active site, will prevent the real substrate from entering and therefore will prevent the enzyme from catalyzing its reaction. The potency of a competitive inhibitor depends on how well it binds to the active site.
Shown below is an example of a competitive inhibitor for an enzyme called isopentenyl diphosphate isomerase (we'll study the reaction it catalyzes in section 15.3A). Looking at the structure of the inhibitor, you can see its resemblance to both the substrate and the product of the reaction - but you can also see that, with its positive charge, it more closely resembles the cationic reaction intermediate.

This makes perfect sense - remember that enzymes have evolved to bind to the transition state of their reactions more tightly than to either the substrate or the product, and by Hammond's postulate we know that the intermediate resembles the transition state more than the substrate or product do. Therefore, an organic chemist who wants to design a potent competitive inhibitor would be wise to try to imitate the structure of the reaction intermediate.
A irreversible inhibitor (also known as a suicide substrate) is also designed to be recognized by its target enzyme, but there is one critical difference. A competitive inhibitor is free to diffuse in and out of the active site (just like an actual enzymatic substrate or product). A covalent inhibitor, in contrast, will form a strong covalent bond to an active site residue after entering the site: once it is in, it will never come out, and thus it has irreversible inactivated the enzyme. Shown below is a covalent inhibitor for isopentenyl diphosphate isomerase:
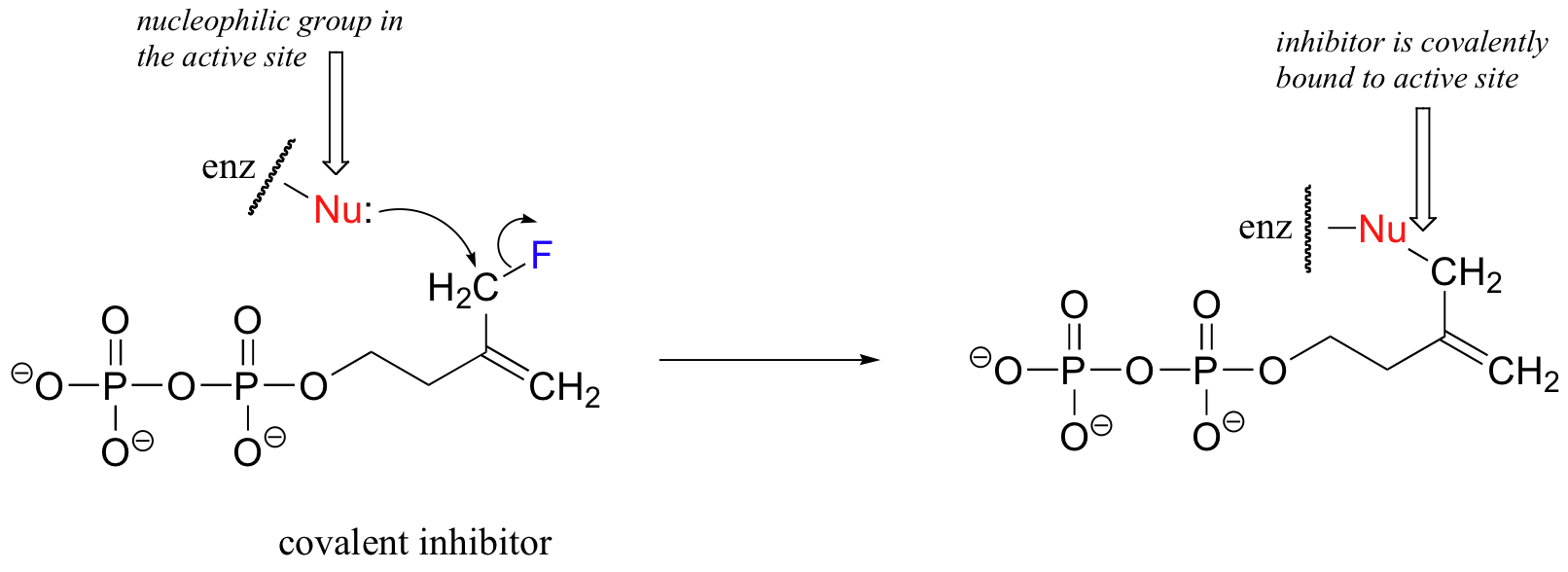
As you can see, the inhibitor resembles the natural substrate, but has a fluorine leaving group on its methyl carbon. The methyl carbon is now an electrophilic 'bait' for any nucleophilic group that happens to be nearby in the active site, and the result is irreversible, covalent inactivation of the enzyme - the inhibitor is locked inside the active sight for good. (Both of these inhibitor examples were first described in Biochemistry 1988, 27, 7315.)
Many drugs work by inhibiting enzymes. The antibiotic penicillin, for example, is an irreversible inhibitor of a bacterial enzyme involved in the construction of cell walls (we will study the action of penicillin in more detail in section 12.5C). The 'statin' family of cholesterol-lowering drugs are all competitive inhibitors of an enzyme called HMG-CoA reductase (section 16.4D), which catalyzes a chemical step in the synthesis of cholesterol.
6.5E: Catalysts in the laboratory
Although we are focusing here on biological catalysis, the role of catalysis in chemistry is by no means limited to enzymes. Chemists synthesizing organic molecules in the laboratory have a wide and ever-growing arsenal of non-biological catalytic strategies at their disposal, a few of which we will see in various places in this text. Often, catalysis in the lab is as simple as adding acid or base to the reaction mixture, in situations where changing the protonation state of a functional group leads to a faster reaction. Metals are frequently used in catalytic roles: one such example that we will discuss later (section 16.5D) is the addition of hydrogen to the double bond of an alkene, a reaction that takes place on the catalytic surface of a metal such as platinum or palladium.
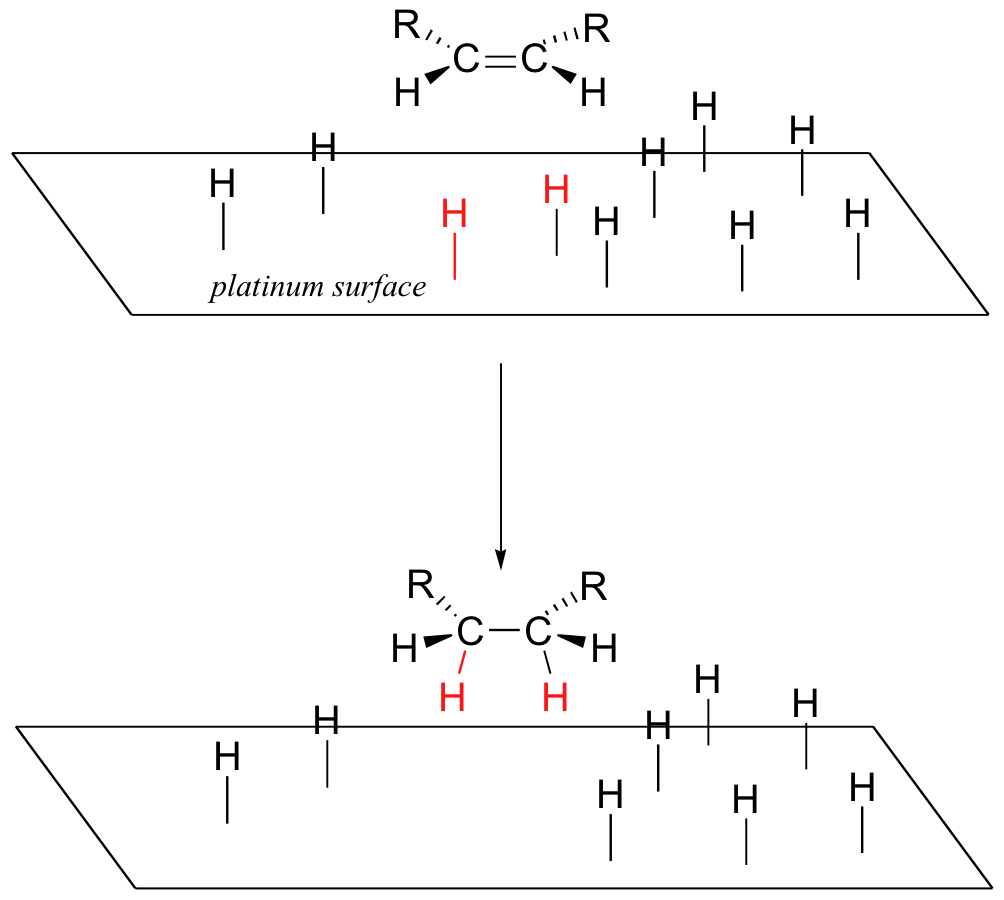
When it comes to efficiency and specificity of catalysis, enzymes for the most part still far outperform their human-designed laboratory counterparts, but most enzymes are inherently fragile and not very effective outside of an aqueous, pH-neutral, 37˚ environment. In addition, an enzyme catalyzes the specific reaction that it has evolved to catalyze, which often is different from the reaction a human chemist wants to carry out. For these reasons, the design and synthesis of large catalytic molecules that mimic the architecture of enzyme active sites has become a very active research field in recent years. Researchers are also exploring ways to use robust, heat-stable enzymes from 'extremophilic' microbes (such as those that live in the boiling water of hot springs) as laboratory catalysts, and are also trying to selectively change the substrate or product specificity of enzymes through the use of site-directed mutagenesis and other genetic engineering techniques.


