7.6: Ácidos polipróticos
- Page ID
- 2365
Polyprotic acids are capable of donating more than one proton. The most important polyprotic acid group from a biological standpoint is the triprotic phosphoric acid. Because phosphoric acid has three acidic protons, it also has three pKa values.
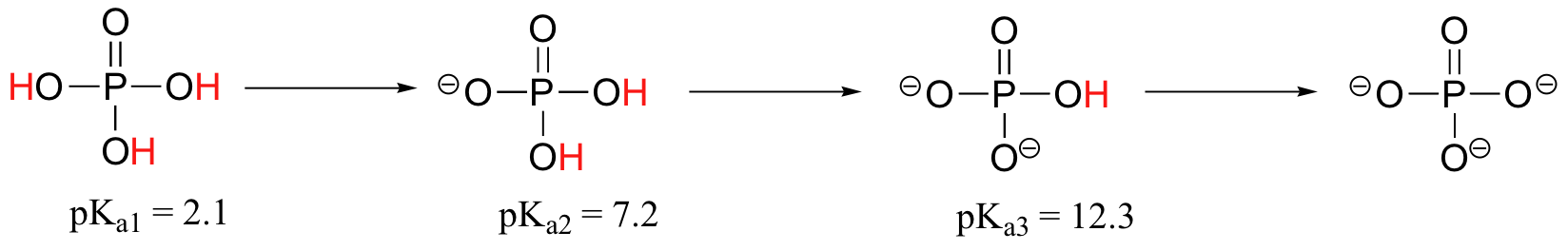
The pKa values for any polyprotic acid always get progressively higher, because it becomes increasingly difficult to stabilize the additional electron density that results from each successive proton donation. H3PO4 is a strong acid because the (single) negative charge on its conjugate base H2PO4- can be delocalized over two oxygen atoms. H2PO4- is substantially less acidic, because proton donation now results in the formation of an additional negative charge, and a –2 charge is inherently higher in energy than a –1 charge, due to negative-negative electrostatic repulsion. The third deprotonation, with formation of a third negative charge, is harder still. We will have more to say about the acidity of phosphate groups in chapter 10.
Free amino acids are polyprotic, with pKa values of approximately 2 for the carboxylic acid group and 9-10 for the amino group. Alanine, for example, has the acid constants pKa1 = 2.3 and pKa2 = 9.9.

Alanine is almost fully protonated and positively charged when dissolved in a solution that is buffered to pH 0.5. At pH 7 , alanine has lost one proton from the carboxylic acid group, and thus is has both a negative and a positive charged group – it is now referred to as a zwitterion. At pH levels above 12, the ammonium group is almost fully deprotonated, and alanine has a negative overall charge.
Some amino acids (arginine, lysine, aspartate, glutamate, tyrosine, and histidine) are triprotic, with a third pKa value associated with the ionizable functional group on the side chain.
Many biological organic molecules have several potentially ionizable functional groups and thus can be considered polyprotic acids. Citric acid, found in abundance in oranges, lemons, and other citrus fruits, has three carboxylic acid groups and pKa values of 3.1, 4.8, and 6.4.

Careful consideration of the structure of citric acid can allow us to predict that it is the middle acid group that has the lowest pKa. This is because the hydroxyl group also bonded to the middle carbon is electron-withdrawing by induction, and a negative charge associated with a conjugate base will be stabilized to the greatest extent on the middle carboxylate. (Notice that in this case, the oxygen, due to its position in the molecule, is not able to exert an electron-donating resonance effect.)
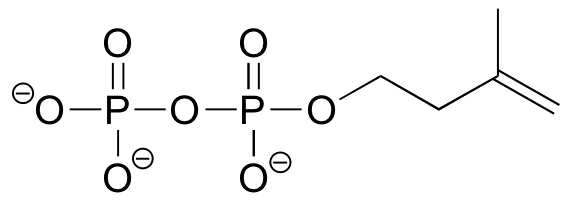
Exercise 7.13: Predict the structure of the organic diphosphate compound below in the protonation state where it has a minus 2 charge.