9.5: Nucleophilic substitution over conjugated pi systems - the SN' mechanism
- Page ID
- 2381
Until now, all of the nucleophilic substitution reactions that we have seen have involved direct displacement at a carbon center: that is, the carbon that is bonded directly to the leaving group is the target for the attacking nucleophile. However, this is not always the case: when the leaving group is bound to a carbon that is part of a system of conjugated π bonds, nucleophilic attack can occur at more than one site. Consider a generalized electrophilic species with a leaving group (X) in the allylic position. In an SN1 mechanism, the carbocation intermediate would have two possible resonance forms.
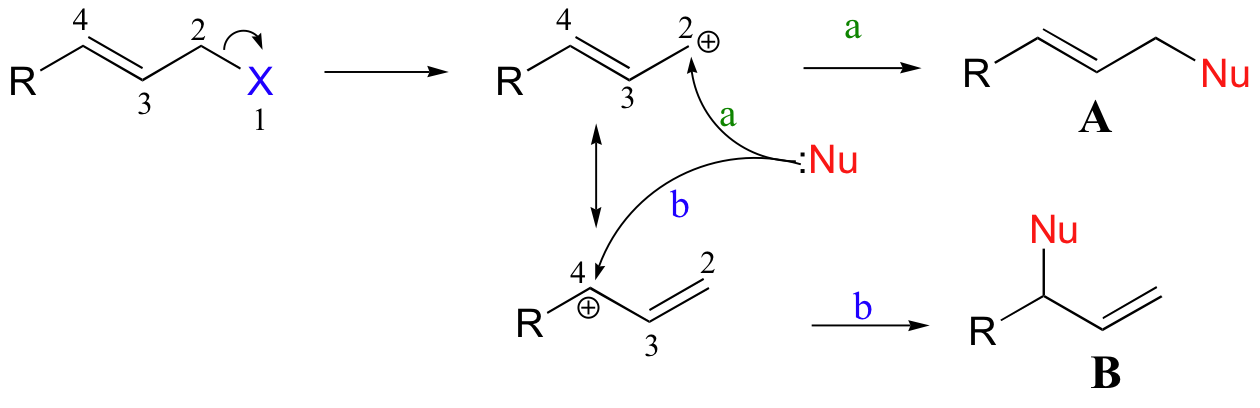
The incoming nucleophile, therefore, could attack either carbon 2 (the carbon originally bonded to the leaving group), or alternatively at carbon 4. These two events would lead to two different substitution products A and B, respectively.
The same can be said of an associative mechanism that does not involve a carbocation intermediate: in this case, however, the concerted mechanisms would be depicted as shown below:

The mechanisms leading to product B are referred to as SN', or nucleophilic allylic substitution. They are also sometimes referred to as 1,4 substitutions, according to a numbering system where the leaving group is designated atom #1.
If the conjugated π orbital system extends further, more SN' products can result: in the following example, 1,6 as well as 1,4 and 1,2 substitutions are possible.
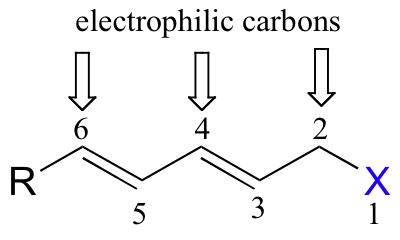
An important example of an enzymatic SN2' reaction is the first step of the reaction catalyzed by anthranilate synthase, an enzyme involved in tryptophan biosynthesis.
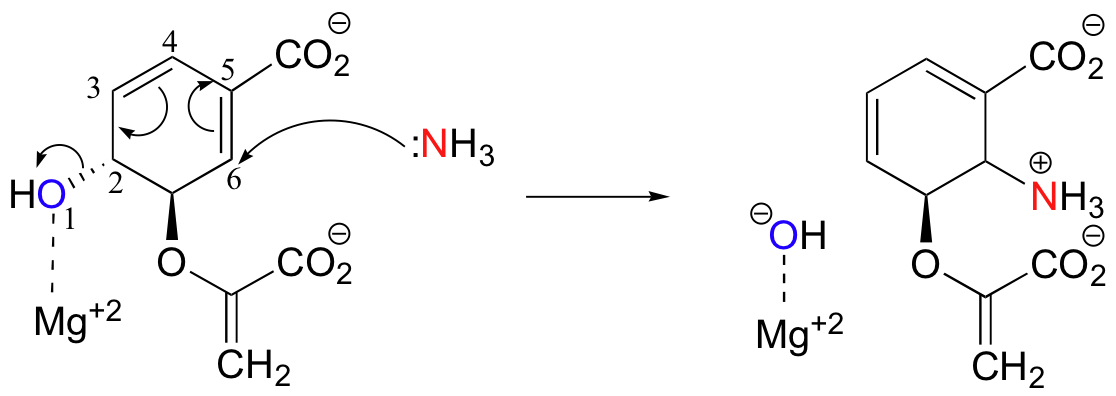
Nucleophilic ammonia attacks in a 1,6 fashion, displacing a hydroxide on the other side of the ring. Note that a hydroxide ion is normally a very poor leaving group. However, in this case the negative charge is stabilized by coordination to a magnesium ion (a Lewis acid) bound in the active site.
In the biosynthesis of morphine in opium poppies (Science 1967, 155, 170; J. Biol. Chem 1995, 270, 31091), the following 1,4 SN' ring-closing reaction occurs:
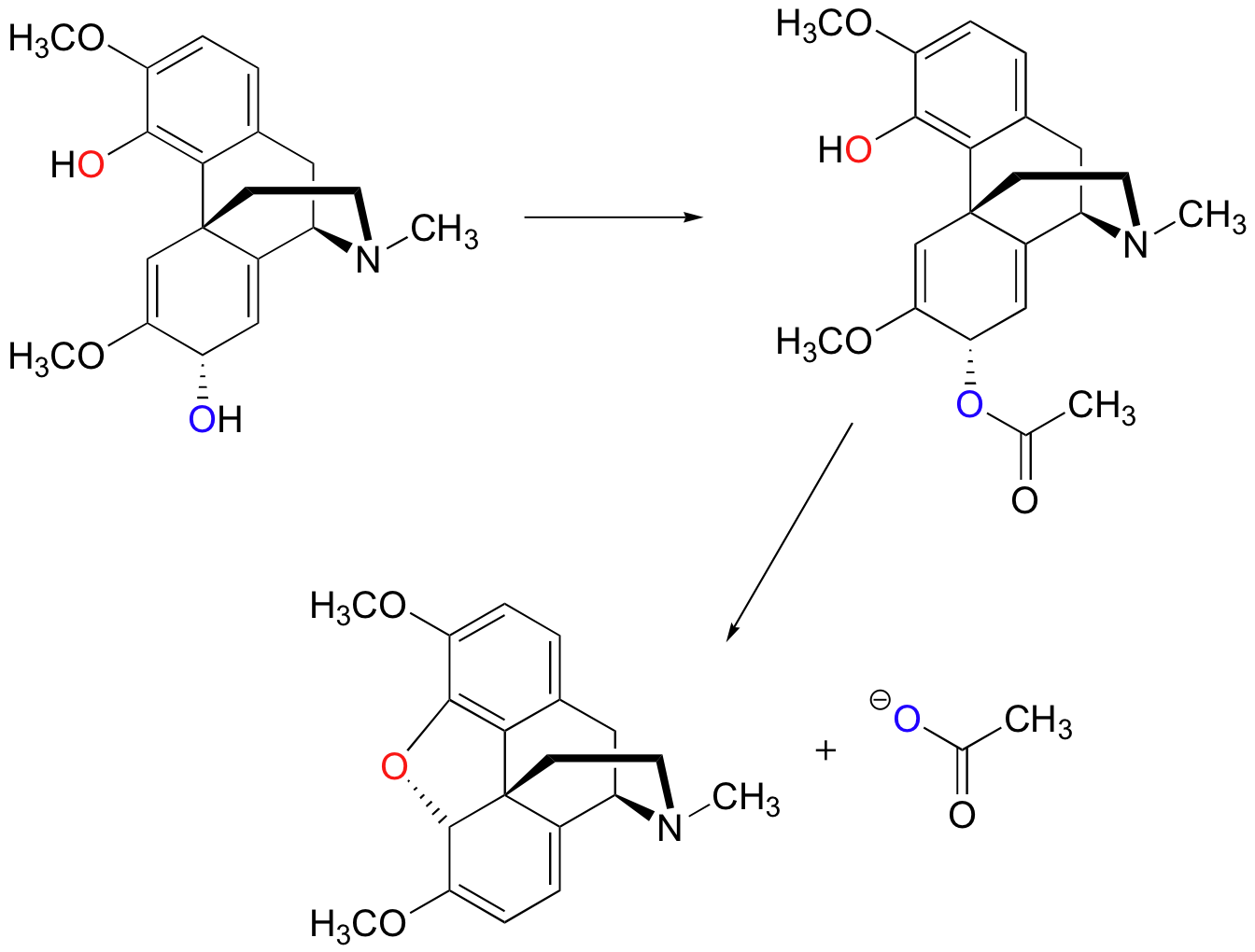
Notice that before the SN' cyclization takes place, the alcohol is first converted to an ester, which is a much better leaving group (we'll study this type of reaction in Chapter 12). Also, notice that the cyclization reaction itself might reasonably be expected to be stepwise (SN1') rather than concerted (SN2'), because the carbocation intermediate formed in a stepwise reaction would be stabilized very well by resonance delocalization of the positive charge:
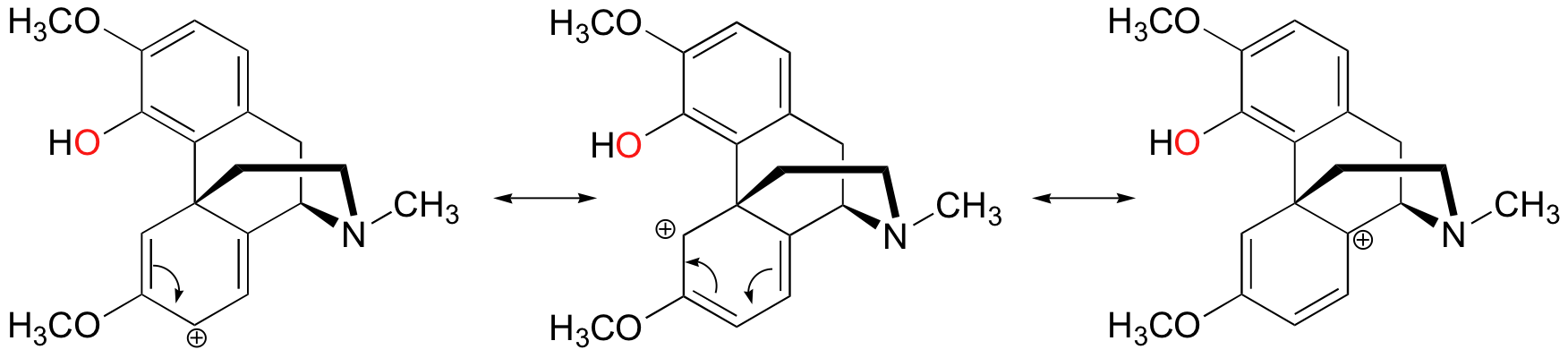
In addition, it is interesting to note that the cyclization stage of the reaction is spontaneous - in other words, it occurs without the need for enzyme catalysis, as soon as the alcohol is converted to an ester (making it a good leaving group).
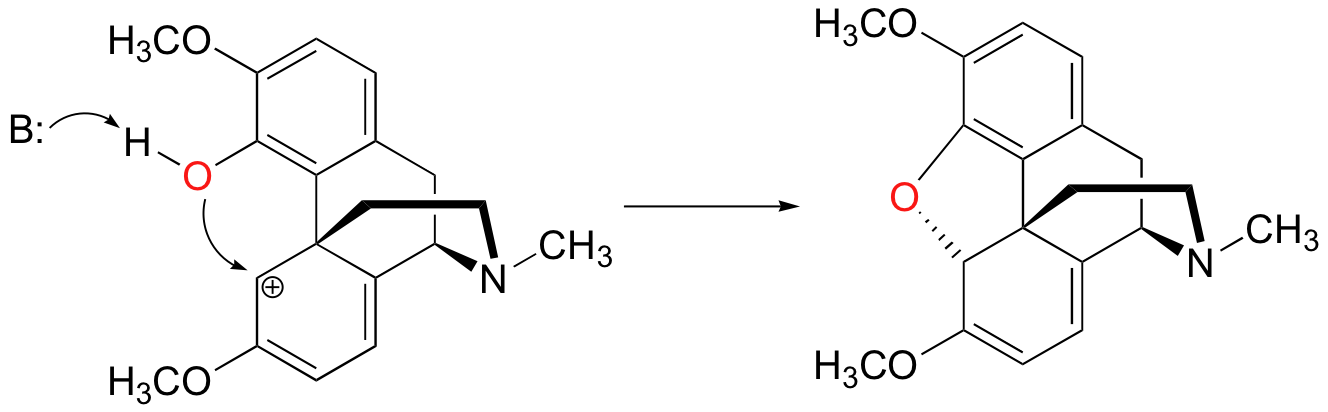
This is possible because the fused-ring structure of the molecule serves to hold the phenol nucleophile in the correct position for attack, thus greatly reducing the entropic component of the energy barrier.


