11.3: La esteroquímica de la reacción de adición nucleófila
- Page ID
- 2392
Notice that in the course of the nucleophilic addition pictured above, the hybridization of the carbonyl carbon changes from sp2 to sp3, meaning that the bond geometry changes from trigonal planar to tetrahedral. It is also important to note that if the starting carbonyl is asymmetric (in other words, if the two R groups are not equivalent), then a new stereocenter has been created. The configuration of the new stereocenter depends upon which side of the carbonyl plane the nucleophile attacks from.

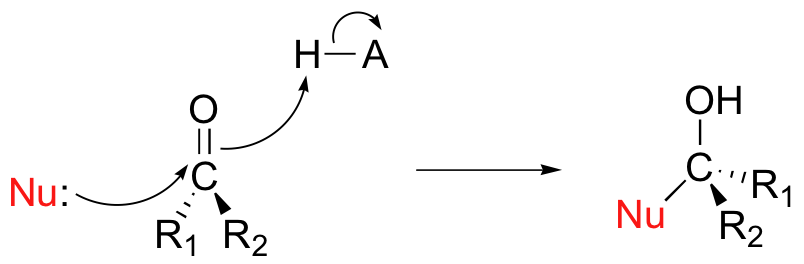
If the reaction is catalyzed by an enzyme, the stereochemistry of addition is tightly controlled, and leads to one specific stereoisomer - this is because the nucleophilic and electrophilic substrates are bound in a specific positions within the active site, so that attack must occur specifically from one side. If, however, the reaction occurs uncatalyzed in solution, then either side of the carbonyl is equally likely to be attacked, and the result will be a 50:50 racemic mixture.
This is the rule for most nonenzymatic reactions, but as with most rules, there are exceptions. If, for example, the geometry of the carbonyl-containing molecule is constrained in such a way that approach by the nucleophile is less hindered from one side, a 50:50 racemic mixture will not necessarily result. Consider camphor, the distinctive-smelling compound found in many cosmetics and skin creams.

Upon inspection it is clear that topside attack and bottom side attack by a nucleophile are nonequivalent in terms of steric hindrance. A relatively simple experiment shows that, when the incoming nucleophile is a hydride ion from the common synthetic reducing agent sodium borohydride (a reaction type we will study in a later chapter), the product of bottom side attack predominates by a ratio of about 6 to 1 (see section 16.4D for more details on this experiment). We can infer from this result that approach from the bottom (si) face of the carbonyl in camphor is less hindered.
. . . on to the next section . . .


