16.11: Reacciones de oxygenasa - monooxigenasas dependientes de flavina
- Page ID
- 2449
Thus far in this chapter, we have focused on the redox reactions that we can broadly characterize as hydrogenation and dehydrogenation - the addition or removal of a hydride ion and a proton. Another important class of redox reactions in biological organic chemistry are those catalyzed by oxygenase and reductase enzymes. In these transformations, one or more oxygen atoms is inserted into or removed from (respectively) an organic substrate.
This oxygenase reaction is part of the biosynthetic pathway for the steroid hormone cortisol:

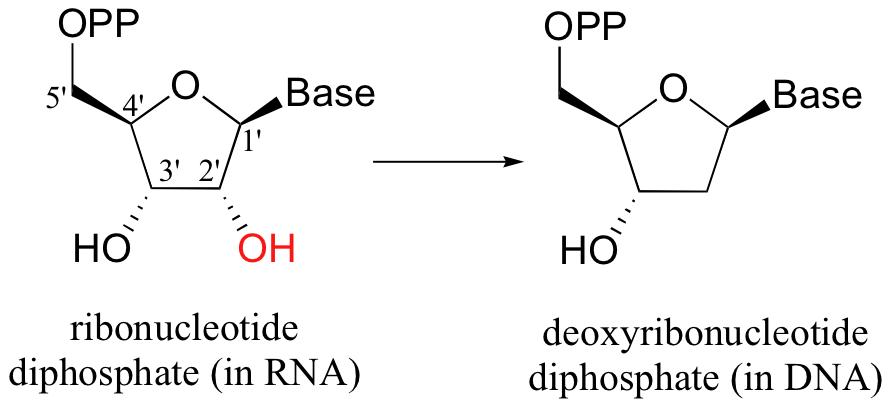
A reductase reaction is required to provide the deoxyribonucleotide component of the DNA backbone:
Many oxygenase and reductase reactions involve enzyme-bound metal centers - iron or copper, for example - and free radical intermediates. For the most part, a mechanistic discussion of these reactions is beyond the scope of this text. However, in this section we will take a quick look at a couple of metal-free monooxygenase reactions in which flavin plays a key role (a monooxygenase inserts a single oxygen atom into its organic substrate, while a dioxygenase inserts two). Later, in section 17.3, we will look two more redox reactions, one a monooxygenase and one a reductase, that involve single-electron (free radical) mechanistic steps.
The idea that some oxygenases are dependent upon the participation of FADH2 may seem surprising at first, given how FADH2 acts as a reducing agent in the alkene hydrogenation reactions discussed in section 16.5A. It turns out that in monooxygenase reactions, the job of FADH2 is to reduce O2 to peroxide in order to activate one of the oxygen atoms for insertion into the organic substrate. The flavin coenzyme reacts with molecular oxygen to form an intermediate species called flavin hydroperoxide.

The mechanism for this step is though in most cases to involve single-electron chemistry and free radical intermediates – we will discuss the single-electron reactions of flavin in section 17.3C, and you will be invited to propose a mechanism for flavin hydroperoxide formation in problem P17.11.
The 'outer' oxygen of flavin hydroperoxide is a potent electrophilic target for attack by the π electrons in an alkene or aromatic group. The epoxide group in 2,3-oxidosqualene, a key intermediate in cholestesterol synthesis, (section 15.7B) is formed by what is essentially an electrophilic addition of oxygen to the C2-C3 double bond of squalene.
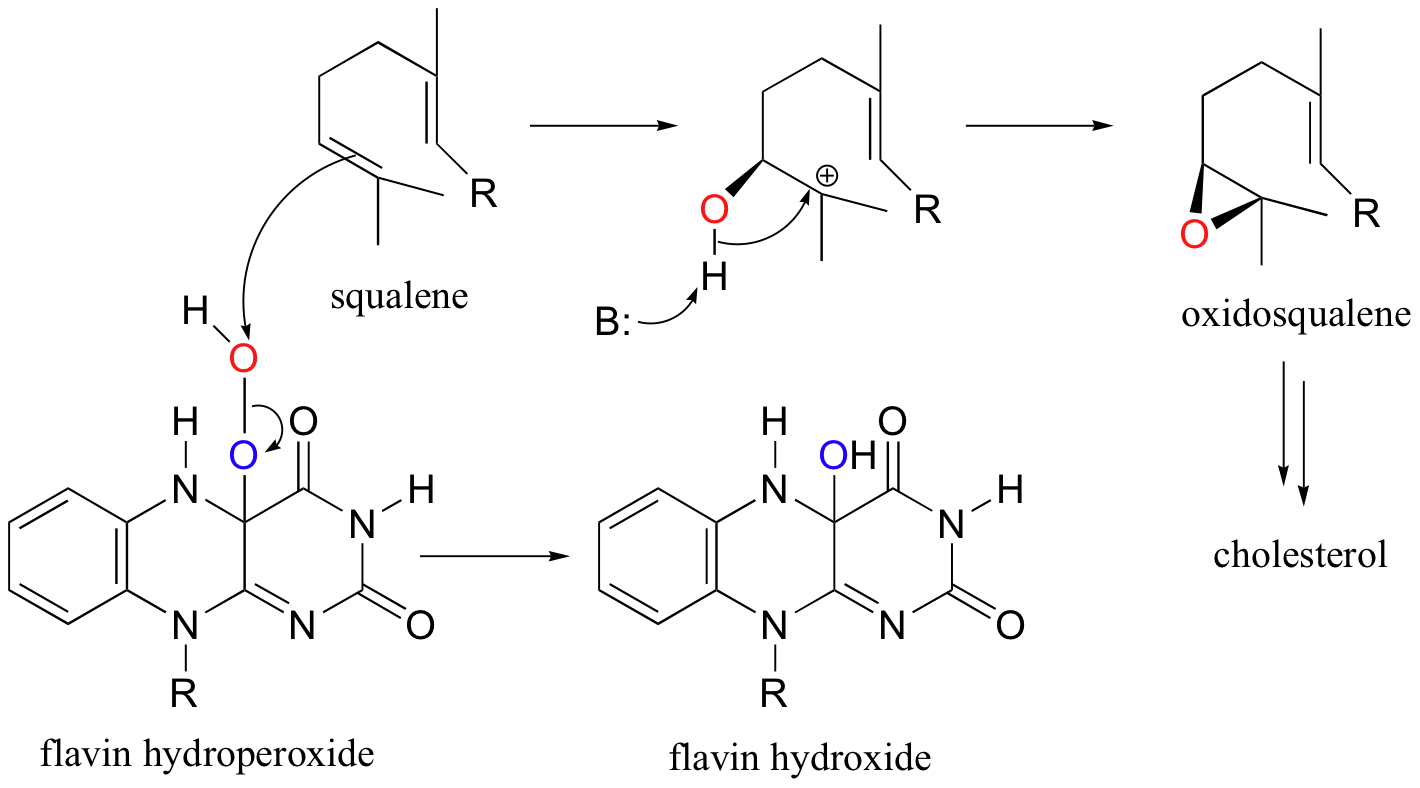
The flavin hydroxide produced in this reaction is converted to FAD via a dehydrogenation reaction, then the catalytic cycle is completed when FAD is reduced back to FADH2 by NADPH.
In the oxidation of tryptophan, an intermediate compound called kynurenine is hydroxylated in what is essentially an electrophilic aromatic substitution (section 15.5) with the reactive oxygen of flavin hydroperoxide acting as the electrophile.



