13.5 Calculaciones de equilibrio
- Page ID
- 1907
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Habilidades Para Desarrollar
- Escriba ecuaciones que representen cambios en la concentración y presión para especies químicas en sistemas de equilibrio.
- Use álgebra para hacer varios tipos de cálculos de equilibrio
Sabemos que en el equilibrio, el valor del cociente de reacción de cualquier reacción es igual a su constante de equilibrio. Por lo tanto, podemos usar la expresión matemática para Q para determinar un número de cantidades asociadas con una reacción en equilibrio o acercándose al equilibrio. Mientras hemos aprendido a identificar en qué dirección se desplazará una reacción para alcanzar el equilibrio, queremos extender esa comprensión a los cálculos cuantitativos. Lo hacemos por evaluando las formas en que las concentraciones de productos y reactivos cambian a medida que la reacción se acerca al equilibrio, teniendo en cuenta las relaciones estequiométricas de la reacción. Este enfoque algebraico para los cálculos de equilibrio se explorará en esta sección.
Los cambios en las concentraciones o presiones de los reactivos y productos ocurren cuando un sistema de reacción se acerca al equilibrio. En esta sección veremos que podemos relacionar estos cambios entre sí usando los coeficientes en la ecuación química balanceada que describe el sistema. Usamos la descomposición del amoníaco como ejemplo.
Al calentar, el amoníaco se descompone reversiblemente en el nitrógeno e el hidrógeno de acuerdo con esta ecuación:
\[\ce{2NH3}(g)⇌\ce{N2}(g)+\ce{3H2}(g)\]
Si una muestra de amoníaco se descompone en un sistema cerrado y la concentración de N2 aumenta por 0.11 M, el cambio en la concentración de N2, Δ[N2], la concentración final menos la concentración inicial, es 0.11 M. El cambio es positivo porque la concentración de N2 aumenta.
El cambio en la concentración de H2, Δ [H2], también es positivo: la concentración de H2 aumenta a medida que el amoníaco se descompone. La ecuación química nos dice que el cambio en la concentración de H2 es tres veces el cambio en la concentración de N2 porque por cada mol de N2 producido, se producen 3 moles de H2.
\[ \begin{align*} \ce{Δ[H2]} &=3×\ce{Δ[N2]} \\[5pt] &=3×(0.11\:M) \\[5pt] &=0.33\:M \end{align*}\]
El cambio en la concentración de NH3, Δ[NH3], es el doble que el de Δ[N2]; la ecuación indica que 2 moles de NH3 se deben descomponer por cada mol de N2 formado. Sin embargo, el cambio en la concentración de NH3 es negativo porque la concentración de amoníaco disminuye cuando se descompone.
\[ \begin{align*} Δ[\ce{NH3}] &=−2×Δ[\ce{N2}] \\[5pt] &=−2×(0.11\:M) \\[5pt] &=−0.22\:M \end{align*}\]
Podemos relacionar estas relaciones directamente con los coeficientes de la ecuación.
&\phantom{Δ[NH3}\ce{2NH3}(g)
&&⇌
&&\phantom{Δ[N2}\ce{N2}(g)
&&+
&&\phantom{Δ[H2]}\ce{3H2}(g)\\
&Δ[\ce{NH3}]=−2×Δ[\ce{N2}]&& && Δ[\ce{N2}]=0.11\:M && && Δ[\ce{H2}]=3×Δ[\ce{N2}]
\end{align}\]
Tenga en cuenta que todos los cambios en un lado de las flechas son del mismo signo y que todos los cambios en el otro lado de las flechas son del signo opuesto.
Si no supiéramos la magnitud del cambio en la concentración de N2, podríamos representarlo con el símbolo x.
Los cambios en las otras concentraciones se representarían como:
\(Δ[\ce{H2}]=3×Δ[\ce{N2}]=3x\)
Los coeficientes en los términos Δ son idénticos a los de la ecuación balanceada para la reacción.
\(\begin{alignat}{3}
&\ce{2NH3}(g)⇌\:&&\ce{N2}(g)+\:&&\ce{3H2}(g)\\
&−2x &&x &&3x
\end{alignat}\)
La forma más sencilla de encontrar los coeficientes para los cambios de concentración en cualquier reacción es usando los coeficientes en la ecuación química equilibrada. El signo del coeficiente es positivo cuando aumenta la concentración pero es negativo cuando la concentración disminuye.
Ejemplo \(\PageIndex{1}\): DETERMINACIÓN DE LOS CAMBIOS RELATIVOS EN LA CONCENTRACIÓN
Complete los cambios en las concentraciones para cada una de las siguientes reacciones.
(a) \(\begin{alignat}{3}
&\ce{C2H2}(g)+\:&&\ce{2Br2}(g)⇌\:&&\ce{C2H2Br4}(g)\\
&x &&\underline{\hspace{40px}} &&\underline{\hspace{40px}}
\end{alignat}\)
(b) \(\begin{alignat}{3}
&\ce{I2}(aq)+\:&&\ce{I-}(aq)⇌\:&&\ce{I3-}(aq)\\
&\underline{\hspace{40px}} &&\underline{\hspace{40px}} &&x
\end{alignat}\)
(c) \(\begin{alignat}{3}
&\ce{C3H8}(g)+\:&&\ce{5O2}(g)⇌\:&&\ce{3CO2}(g)+\:&&\ce{4H2O}(g)\\
&x &&\underline{\hspace{40px}} &&\underline{\hspace{40px}} &&\underline{\hspace{40px}}
\end{alignat}\)
Solución
(a) \(\begin{alignat}{3}
&\ce{C2H2}(g)+\:&&\ce{2Br2}(g)⇌\:&&\ce{C2H2Br4}(g)\\
&x &&2x &&-x
\end{alignat}\)
(b) \(\begin{alignat}{3}
&\ce{I2}(aq)+\:&&\ce{I-}(aq)⇌\:&&\ce{I3-}(aq)\\
&-x &&-x &&x
\end{alignat}\)
(c) \(\begin{alignat}{3}
&\ce{C3H8}(g)+\:&&\ce{5O2}(g)⇌\:&&\ce{3CO2}(g)+\:&&\ce{4H2O}(g)\\
&x &&5x &&-3x &&-4x
\end{alignat}\)
Ejercicio \(\PageIndex{1}\)
Complete los cambios en las concentraciones para cada una de las siguientes reacciones.
(a) \(\begin{alignat}{3}
&\ce{2SO2}(g)+\:&&\ce{O2}(g)⇌\:&&\ce{2SO3}(g)\\
&\underline{\hspace{40px}} &&x &&\underline{\hspace{40px}}
\end{alignat}\)
(b) \(\begin{alignat}{3}
&\ce{C4H8}(g)⇌\:&&\ce{2C2H4}(g)\\
&\underline{\hspace{40px}} &&-2x
\end{alignat}\)
(c) \(\begin{alignat}{3}
&\ce{4NH3}(g)+\:&&\ce{7H2O}(g)⇌\:&&\ce{4NO2}(g)+\:&&\ce{6H2O}(g)\\
&\underline{\hspace{40px}} &&\underline{\hspace{40px}} &&\underline{\hspace{40px}} &&\underline{\hspace{40px}}
\end{alignat}\)
- Respuesta a
-
2x, x, −2x
- Respuesta b
-
x, −2x
- Respuesta c
-
4x, 7x, −4x, −6x or −4x, −7x, 4x, 6x
Cálculos que involucran las concentraciones de equilibrio
Debido a que el valor del cociente de la reacción de cualquier reacción en equilibrio es igual a su constante de equilibrio, podemos usar la expresión matemática para Qc (es decir, la ley de acción de masas) para determinar una cantidad de cantidades asociadas con una reacción en equilibrio. Puede ayudar si tenemos en cuenta que Qc=Kc (en equilibrio) en todas estas situaciones y que solo hay tres tipos básicos de cálculos:
- Cálculo de un constante de equilibrio. Si se saben las concentraciones de los reactivos y los productos en equilibrio, se puede calcular el valor del constante de equilibrio para la reacción.
- Cálculo de las concentraciones de equilibrio faltantes. Si se sabe el valor del constante de equilibrio y todas las concentraciones de equilibrio, excepto una, se puede calcular la concentración no dada.
- Cálculo de las concentraciones de equilibrio a partir de las concentraciones iniciales. Si se sabe el valor del constante de equilibrio y un conjunto de concentraciones de los reactivos y los productos que no están en equilibrio, se pueden calcular las concentraciones en equilibrio.
Se podría producir una lista similar usando QP, KP y presión parcial. Miraremos la resolución de cada uno de estos casos en secuencia.
Cálculo del constante de equilibrio
Dado que la ley de acción de masas es la única ecuación que tenemos para describirEl ejemplo \(\PageIndex{1}\) nos mostró cómo determinar el constante de equilibrio de una reacción si sabemos las concentraciones de los reactivos y los productos en equilibrio. El siguiente ejemplo muestra cómo usar la estequiometría de la reacción y una combinación de concentraciones iniciales y concentraciones de equilibrio para determinar un constante de equilibrio. Esta técnica, comúnmente llamada la tabla de ICE (inicial, cambio y equilibrio) será útil para resolver muchos problemas de equilibrio. Se hace un gráfico que comienza con la reacción de equilibrio en cuestión. Debajo de la reacción se enumeran las concentraciones iniciales de los reactivos y los productos: estas condiciones generalmente se proporcionan en el problema y consideramos que no ha ocurrido ningún cambio en el equilibrio. La siguiente fila del gráfico es el cambio que ocurre cuando el sistema se desplaza hacia el equilibrio; no olvide considerar la estequiometría de la reacción como se describe en una sección anterior de este capítulo. La última fila contiene las concentraciones una vez que se ha alcanzado el equilibrio.
El ejemplo \(\PageIndex{1}\) nos mostró cómo determinar el constante de equilibrio de una reacción si sabemos las concentraciones de los reactivos y los productos en equilibrio. El siguiente ejemplo muestra cómo usar la estequiometría de la reacción y una combinación de concentraciones iniciales y concentraciones de equilibrio para determinar un constante de equilibrio. Esta técnica, comúnmente llamada la tabla de ICE (inicial, cambio y equilibrio) será útil para resolver muchos problemas de equilibrio. Se hace un gráfico que comienza con la reacción de equilibrio en cuestión. Debajo de la reacción se enumeran las concentraciones iniciales de los reactivos y los productos: estas condiciones generalmente se proporcionan en el problema y consideramos que no ha ocurrido ningún cambio en el equilibrio. La siguiente fila del gráfico es el cambio que ocurre cuando el sistema se desplaza hacia el equilibrio; no olvide considerar la estequiometría de la reacción como se describe en una sección anterior de este capítulo. La última fila contiene las concentraciones una vez que se ha alcanzado el equilibrio.
Ejemplo \(\PageIndex{2}\): CÁLCULO DE UN CONSTANTE DE EQUILIBRIO
Las moléculas de yodo reaccionan de manera reversible con los iones de yoduro para producir los iones de yoduro.
\[\ce{I2}(aq)+\ce{I-}(aq)⇌\ce{I3-}(aq)\]
Si una solución con las concentraciones de I2 e I− ambas iguales a 1.000 × 10−3 M antes de que la reacción empiece da una concentración de equilibrio de I2 de 6.61×10−4 M, ¿Cuál es el constante de equilibrio para la reacción?
Solución
Comenzaremos este problema calculando los cambios en la concentración cuando la reacción se acerca hacia el equilibrio. Luego determinamos las concentraciones de equilibrio y, finalmente, el constante de equilibrio. Primero, configuramos una tabla con las concentraciones iniciales, los cambios en las concentraciones y las concentraciones de equilibrio usando −x como el cambio en la concentración de I2.
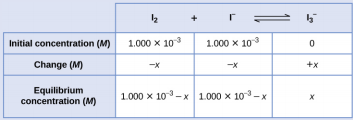
Dado que se da la concentración de equilibrio de I2, podemos resolver para x. En equilibrio, la concentración de I2 es 6.61 × 10−4 M, para que
\(1.000×10^{−3}−x=6.61×10^{−4}\)
\(x=1.000×10^{−3}−6.61×10^{−4}\)
\(=3.39×10^{−4}\:M\)
Ahora podemos completar la tabla con las concentraciones en equilibrio

Ahora calculamos el valor del constante de equilibrio.
\(K_c=Q_c=\ce{\dfrac{[I3- ]}{[I2][I- ]}}\)
\(=\dfrac{3.39×10^{−4}\:M}{(6.61×10^{−4}\:M)(6.61×10^{−4}\:M)}=776\)
Ejercicio \(\PageIndex{2}\)
El etanol y el ácido acético reaccionan para formar el agua y el acetato de etilo, el solvente que es responsable para el olor de algunos quitaesmaltes.
\(\ce{C2H5OH + CH3CO2H ⇌ CH3CO2C2H5 + H2O}\)
Cuando 1 mol de C2H5OH y 1 mol de CH3CO2H reaccionan en 1 L del solvente dioxano, el equilibrio se establece cuando \(\dfrac{1}{3}\) mol de cada uno de los reactivos permanece. Calcule el constante de equilibrio para la reacción. (Nota: el agua no es un disolvente en esta reacción).
- Respuesta
-
Kc = 4
Cálculo de una concentración de equilibrio faltante
Si sabemos el constante de equilibrio para una reacción y sabemos las concentraciones en el equilibrio de todos los reactivos y los productos, excepto uno, podemos calcular la concentración que falta.
Ejemplo \(\PageIndex{3}\): Calculation of a Missing Equilibrium Concentration
Los óxidos de nitrógeno son contaminantes del aire producidos por la reacción del nitrógeno y el oxígeno a altas temperaturas. A 2000 °C, el valor del constante de equilibrio para la reacción, \(\ce{N2}(g)+\ce{O2}(g) ⇌ \ce{2NO}(g)\), es 4.1 × 10−4. Encuentre la concentración de NO(g) en una mezcla de equilibrio con aire a una presión de 1 atm a esta temperatura. En el aire, [N2] = 0.036 mol/L y [O2] 0.0089 mol/L.
Solución
Se nos dan todas las concentraciones de equilibrio excepto la de NO. Por lo tanto, podemos resolver la concentración del equilibrio faltante si reorganizamos la ecuación para resolver por el constante de equilibrio.
\(K_c=Q_c=\ce{\dfrac{[NO]^2}{[N2][O2]}}\)
\(\ce{[NO]^2}=K_c\ce{[N2][O2]}\)
\(\ce{[NO]}=\sqrt{K_c\ce{[N2][O2]}}\)
\(=\sqrt{(4.1×10^{−4})(0.036)(0.0089)}\)
\(=\sqrt{1.31×10^{−7}}\)
\(=3.6×10^{−4}\)
Así [NO] es 3.6 × 10−4 mol/L en equilibrio bajo estas condiciones.
Podemos verificar nuestra respuesta por sustituyendo todas las concentraciones de equilibrio en la expresión para el cociente de la reacción para ver si es igual al constante de equilibrio.
La respuesta fue verificada; nuestro valor calculado da el constante de equilibrio dentro del error asociado con las cifras significativas en el problema.
Ejercicio \(\PageIndex{3}\)
El constante de equilibrio para la reacción del nitrógeno e el hidrógeno para producir el amoníaco a cierta temperatura es 6.00 × 10−2. Calcule la concentración de equilibrio de amoníaco si las concentraciones de equilibrio del nitrógeno e el hidrógeno son 4.26 M y 2.09 M, respectivamente.
- Respuesta
-
1.53 mol/L
Cálculo de los cambios en la concentración
Si sabemos el constante de equilibrio para una reacción y un conjunto de concentraciones de reactivos y productos que no están en equilibrio, podemos calcular los cambios en las concentraciones a medida que el sistema llega al equilibrio, así como las nuevas concentraciones cuando la reacción está en equilibrio. El procedimiento típico se puede resumir en cuatro pasos.
1. Determine la dirección en la cual la reacción procede al equilibrio.
- Escriba una ecuación química equilibrada para la reacción.
- Si la dirección en la cual la reacción debe proceder para alcanzar el equilibrio no es obvia, calcule Qc a partir de las concentraciones iniciales y compárelo con Kc para determinar la dirección del cambio.
2. Determine los cambios relativos necesarios para alcanzar el equilibrio, luego escriba las concentraciones de equilibrio en términos de estos cambios.
- Defina los cambios en las concentraciones iniciales que se necesitan para que la reacción alcance el equilibrio. Generalmente, representamos el cambio más pequeño con el símbolo x y expresamos los otros cambios en términos del cambio más pequeño.
- Defina las concentraciones de equilibrio que faltan en términos de las concentraciones iniciales y los cambios en la concentración determinados en (a).
3. Resuelva para tomar en cuenta el cambio y las concentraciones de equilibrio.
- Sustituya las concentraciones de equilibrio en la expresión del constante de equilibrio, resuelva para x y verifique cualquier suposición usada para encontrar x.
- Calcule las concentraciones de equilibrio.
4. Verifique la aritmética.
- Verifique las concentraciones de equilibrio calculadas sustituyéndolas en la expresión de equilibrio y determinando si dan el constante de equilibrio.
A veces, un paso en particular puede diferir de un problema a otro; puede ser más complejo en algunos problemas y menos complejo en otros. Sin embargo, cada cálculo de las concentraciones de equilibrio a partir de un conjunto de concentraciones iniciales involucrará estos pasos.
Al resolver problemas de equilibrio que involucran cambios en la concentración, a veces es conveniente configurar una tabla ICE, como se describe en la sección anterior.
Ejemplo \(\PageIndex{4}\): CÁLCULO DE LA CONCENTRACIÓN Cuando una reacción alcanza equilibrio
Bajo ciertas condiciones, el constante de equilibro para la descomposición de PCl5(g) en PCl3(g) y Cl2(g) es 0.0211. ¿Cuáles son las concentraciones de equilibrio de PCl5, PCl3, y Cl2 si la concentración inicial de PCl5 fue de 1.00 M?
Solución
Use el proceso paso a paso descrito anteriormente.
1. Determine la dirección en la que la reacción procede.
La ecuación equilibrada para la descomposición de PCl5 es
Debido a que inicialmente no tenemos productos, Qc = 0, la reacción procederá hacia la derecha.
2. Determine los cambios relativos necesarios para alcanzar el equilibrio, luego escriba las concentraciones de equilibrio en términos de estos cambios.
Representemos el aumento en la concentración de PCl3 por el símbolo x. Los otros cambios se pueden escribir en términos de x considerando los coeficientes en la ecuación química.
&\ce{PCl5}(g)⇌\:&&\ce{PCl3}(g)+\:&&\ce{Cl2}(g)\\
&-x &&x &&x
\end{alignat}\)
Los cambios en la concentración y las expresiones para las concentraciones de equilibrio son:

3. Resuelva para el cambio y las concentraciones de equilibrio.
Sustituyendo las concentraciones de equilibrio en la ecuación del constante de equilibrio nos da
Esta ecuación contiene solo una variable, x, el cambio en la concentración. Podemos escribir la ecuación como una ecuación cuadrática y resolver para x usando la fórmula cuadrática.
Una ecuación de la forma ax2 + bx + c = 0 se puede reorganizar para resolver por x
En este caso, a = 1, b = 0.0211 y c = −0.0211. Sustituyendo los valores apropiados para a, b y c produce:
Por eso
o
A veces, las ecuaciones cuadráticas tienen dos soluciones diferentes, una que es físicamente posible y otra que es físicamente imposible (una raíz extraña). En este caso, la segunda solución (−0.156) es físicamente imposible porque sabemos que el cambio debe ser un número positivo (de lo contrario, terminaríamos con valores negativos para las concentraciones de los productos). Por lo tanto, x = 0.135 M.
Las concentraciones de equilibrio son
\(\ce{[PCl3]}=x=0.135\:M\)
\(\ce{[Cl2]}=x=0.135\:M\)
4. Cheque la aritmética
La sustitución en la expresión de Kc (para verificar el cálculo) nos da
El constante de equilibrio calculado de las concentraciones de equilibrio es igual al valor de Kc dado en el problema (cuando se redondea al número apropiado de cifras significativas). Así, las concentraciones de equilibrio calculadas son iguales.
Ejercicio \(\PageIndex{4A}\)
El ácido acético, CH3CO2H, reacciona con el etanol, C2H5OH, para formar el agua y el acetato de etilo, CH3CO2C2H5.
\[\ce{CH3CO2H + C2H5OH ⇌ CH3CO2C2H5 + H2O} \nonumber\]
El constante de equilibrio para esta reacción con el dioxano como disolvente es 4.0. ¿Cuáles son las concentraciones de equilibrio cuando una mezcla que es 0.15 M en CH3CO2H, 0.15 M en C2H5OH, 0.40 M en CH3CO2C2H5 y 0.40 M en H2O se mezclan en suficiente dioxano para obtener 1.0 L de solución?
- Respuesta:
-
[CH3CO2H] = 0.36 M, [C2H5OH] = 0.36 M, [CH3CO2C2H5] = 0.17 M, [H2O] = 0.17 M
Ejercicio \(\PageIndex{4B}\)
Un matraz de 1.00 L se llena con 1.00 mol de H2 y 2.00 moles de I2. El valor del constante de equilibrio para la reacción de hidrógeno y yodo que reacciona para formar el yoduro de hidrógeno es 50.5 en las condiciones dadas. ¿Cuáles son las concentraciones de equilibrio de H2, I2 y HI en moles / L?
\[\ce{H2}(g)+\ce{I2}(g)\rightleftharpoons\ce{2HI}(g) \nonumber\]
- Respuesta
-
[H2] = 0.06 M, [I2] = 1.06 M, [HI] = 1.88 M
A veces es posible usar la intuición química para encontrar soluciones a los problemas de equilibrio sin resolver una ecuación cuadrática (o más complicada). Primero, sin embargo, es útil verificar que el equilibrio se pueda obtener a partir de dos extremos: todos (o principalmente) reactivos y todos (o principalmente) productos.
Considere la ionización de 0.150 M HA, un ácido débil.
La forma más obvia de determinar las concentraciones de equilibrio sería comenzar solo con los reactivos. Este método se podría llamar el punto de partida "todo reactivo". Usando x para la cantidad de ácido ionizado en equilibrio, esta es la tabla ICE y la solución.

Configurando y resolviendo la ecuación cuadrática nos da
Usando la raíz positiva, las concentraciones de equilibrio son
Una forma menos obvia de resolver el problema sería asumir que todos los HA se ionizan primero, luego el sistema llega al equilibrio. Esto se podría llamar el punto de partida de "todos los productos". Asumiendo que todo HA se ioniza nos da
Utilizándolos como las concentraciones iniciales e "y" para representar la concentración de HA en equilibrio, esta es la tabla ICE para este punto de partida.
Conserve algunas cifras significativas adicionales para minimizar los problemas de redondeo.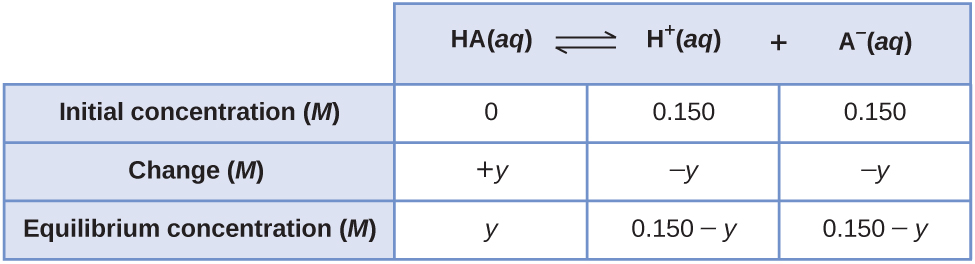
Configurando y resolviendo la ecuación cuadrática nos da
Conserve algunas cifras significativas adicionales para minimizar los problemas de redondeo.
Redondear cada solución a tres cifras significativas nos da
El uso de la raíz físicamente significativa (0.140 M) nos da las concentraciones de equilibrio como
Por lo tanto, los dos métodos nos dan los mismos resultados (a tres decimales) y muestran que ambos puntos de partida producen las mismas condiciones de equilibrio. El punto de partida de "todos los reactivos" resultó en un cambio relativamente pequeño (x) porque el sistema estaba casi en equilibrio, mientras que el punto de partida de "todo el producto" tuvo un cambio relativamente grande (y) que era casi del tamaño de las concentraciones iniciales. Se puede decir que un sistema que comienza a "cerrarse" al alcanza el lequilibrio requerirá solo un cambio "pequeño" en las condiciones (x) para llegar al equilibrio.
Recuerde que un Kc pequeño significa que poco de los reactivos forman productos y un Kc grande significa que la mayoría de los reactivos forman productos. Si el sistema se puede organizar de modo que comience a "cerrarse" al alcanzar el equilibrio, si el cambio (x) es pequeño en comparación con cualquier concentración inicial, se puede descuidar. Generalmente, un error pequeño se define como el resultado de un error de menos del 5%. Los siguientes dos ejemplos demuestran esto.
Ejemplo \(\PageIndex{5}\): SOLUCIÓN APROXIMADA EMPEZANDO CERCA DEL EQUILIBRIO
¿Cuáles son las concentraciones en equilibrio de una solución a 0.15 M de HCN?
Solución usando "x" para representar la concentración de cada producto en equilibrio nos da esta tabla ICE.
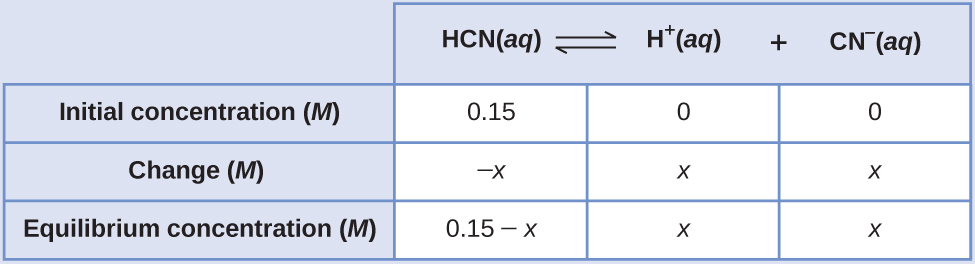
La solución exacta se puede obtener usando la fórmula cuadrática con
resolviendo
\[x^2+4.9×10^{−10}−7.35×10^{−11}=0\]
Así [H+] = [CN–] = x = 8.6 × 10–6 M and [HCN] = 0.15 – x = 0.15 M.
En este caso, nuestra intuición química nos puede dar una solución más simple. A partir del constante de equilibrio y las condiciones iniciales, x debe ser pequeño en comparación con 0.15 M. Más formalmente, si \(x≪0.15\), entonces 0.15 - x ≈ 0.15. Si esta suposición es cierta, entonces se puede simplificar y obtener x
\[K_c=\dfrac{(x)(x)}{0.15−x}≈\dfrac{x^2}{0.15}\]
\[4.9×10^{−10}=\dfrac{x^2}{0.15}\]
\[x^2=(0.15)(4.9×10^{−10})=7.4×10^{−11}\]
\[x=\sqrt{7.4×10^{−11}}=8.6×10^{−6}\:M\]
En este ejemplo, resolviendo la ecuación exacta (cuadrática) y usando aproximaciones nos dio el mismo resultado a dos cifras significativas. Aunque la mayoría de las veces la aproximación es un poco diferente en comparación con la solución exacta, si el error es menos que 5%, la solución aproximada se considera válida. En este problema, el 5% se aplica a IF (0.15 - x) ≈ 0.15 M, entonces si
es menos que 5%, como en este caso, la suposición es válida. La solución aproximada es, por lo tanto, una solución válida.
Ejercicio \(\PageIndex{5}\)
¿Cuáles son las concentraciones de equilibrio en una solución de NH3 a 0.25 M?
Suponga que x es mucho menor que 0.25 M y calcule el error en su suposición.
- Respuesta
-
\(\ce{[OH- ]}=\ce{[NH4+]}=0.0021\:M\); [NH3] = 0.25 M, error = 0.84%
El segundo ejemplo requiere que la información original se procese un poco, pero aún se puede resolver usando una pequeña aproximación de x.
Ejemplo \(\PageIndex{6}\): SOLUCIÓN APROXIMADA DESPUÉS DE UN CAMBIO EN LA CONCENTRACIÓN INICIAL
Los iones de cobre(II) forman un complejo en la presencia del amoníaco.
Si se agregan 0.010 mol de Cu2+ a 1.00 L de una solución que es 1.00 M de NH3, ¿Cuáles son las concentraciones cuando el sistema alcanza el equilibrio?
Solución
La concentración inicial del cobre (II) es 0.010 M. El constante de equilibrio es muy grande, por eso sería mejor comenzar con la mayor cantidad de producto posible porque "todos los productos" están mucho más cerca del equilibrio que "todos los reactivos". Tenga en cuenta que Cu2+ es el reactivo limitante; si todo del cobre (II) a 0.010 M reacciona para formar el producto, las concentraciones serían
\[\ce{[Cu^2+]}=0.010−0.010=0\:M\]
\[\ce{[Cu(NH3)4^2+]}=0.010\:M\]
\[\ce{[NH3]}=1.00−4×0.010=0.96\:M\]
El uso de estos valores "desplazados" como las concentraciones iniciales con x representando la concentración de los iones de cobre(II) libres en equilibrio nos da esta tabla ICE.

Como estamos comenzando cerca del equilibrio, el cambio de x debe ser pequeño para que
Seleccione la concentración más pequeña para realizar la regla del 5%.
El porcentaje es mucho menos del 5%, por lo que los supuestos son válidos. Las concentraciones en equilibrio son
Al comenzar con la cantidad máxima de producto, este sistema estaba cerca del equilibrio y el cambio (x) era muy pequeño. Con solo un pequeño cambio requerido para llegar al equilibrio, la ecuación para x se simplificó enormemente y dio un resultado válido dentro del máximo de error del 5%.
Ejercicio \(\PageIndex{6}\)
¿Cuáles son las concentraciones de equilibrio cuando se agregan 0.25 mol de Ni2+ a 1.00 L de solución de NH3 2.00 M?
\[\ce{Ni^2+}(aq)+\ce{6NH3}(aq)⇌\ce{Ni(NH3)6^2+}(aq) \nonumber \]
con \(K_c=5.5×10^8\).Con un constante de equilibrio tan grande, primero forme la mayor cantidad de producto posible, luego suponga que solo queda una pequeña cantidad (x) del producto. Calcule el error en su suposición.
- Respuesta
-
\(\ce{[Ni(NH3)6^2+]}=0.25\:M\), [NH3] = 0.50 M, [Ni2+] = 2.9 × 10–8 M, error = 1.2 × 10–5%
Resumen
Las relaciones de la velocidad de cambio en las concentraciones de una reacción son iguales a las relaciones de los coeficientes en la ecuación química equilibrada. El signo del coeficiente de X es positivo cuando la concentración aumenta y negativo cuando la concentración disminuye. Aprendimos a abordar tres tipos básicos de problemas de equilibrio. Cuando se dan las concentraciones de los reactivos y de los productos en equilibrio, podemos resolver para el constante de equilibrio; cuando se da el constante de equilibrio y algunas de las concentraciones involucradas, podemos resolver las concentraciones faltantes; y cuando se nos da el constante de equilibrio y las concentraciones iniciales, podemos resolver para las concentraciones en equilibrio.
Contribuyentes
Paul Flowers (Universidad de Carolina del Norte - Pembroke), Klaus Theopold (Universidad de Delaware) y Richard Langley (Stephen F. Austin Universidad del Estado) con autores contribuyentes. Contenido del libro de texto producido por la Universidad de OpenStax tiene licencia de Atribución de Creative Commons Licencia 4.0 licencia. Descarge gratis en http://cnx.org/contents/85abf193-2bd...a7ac8df6@9.110)."
Ana Martinez (amartinez02@saintmarys.edu) contribuyó a la traducción de este texto.


