17.5: Problems for Chapter 17
- Page ID
- 2459
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Link to Solution Manual
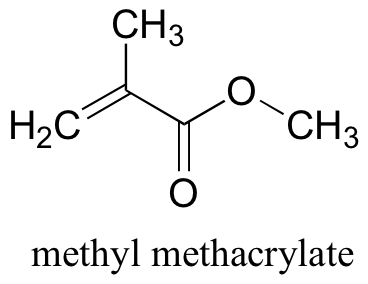
P17.1: Plexiglass is a polymer of methyl methacrylate. Show a mechanism for the first two propagation steps of polymerization (just use X• to denote the radical initiator), and show a structure for the plexiglass polymer.
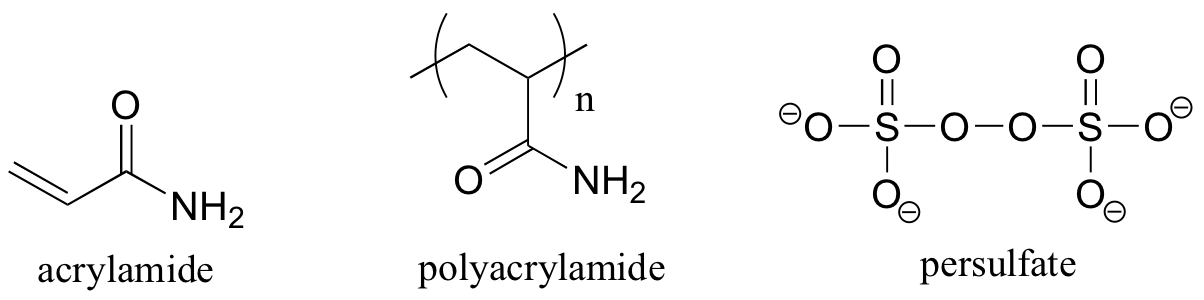
P17.2: A common way to separate proteins in the biochemistry lab is through a technique called polyacrylamide gel electrophoresis (PAGE).
a) Polyacrylamide is formed through radical polymerization of acrylamide monomer, with ammonium persulfate used as the radical initiator. Propose a mechanism for this process.
b) Polyacrylamide by itself does not form a gel - the long polyacrylamide chains simply slip against each other, like boiled spaghetti. To make a real gel, with pores for the proteins to slip through, we need a crosslinker - something to tie the chains together, forming a three-dimensional 'web'. Usually, a small amount of bis-acrylamide is added to the acrylamide solution for this purpose.
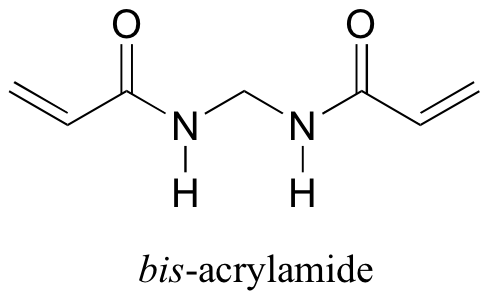
Propose a radical mechanism showing how bisacrylamide might form crosslinks between growing polyacrylamide chains.
P17.3: Resveratrol is a natural antioxidant found in red wine (see section 17.2D). Use resonance structure to illustrate how the resveratrol radical is stabilized by resonance.
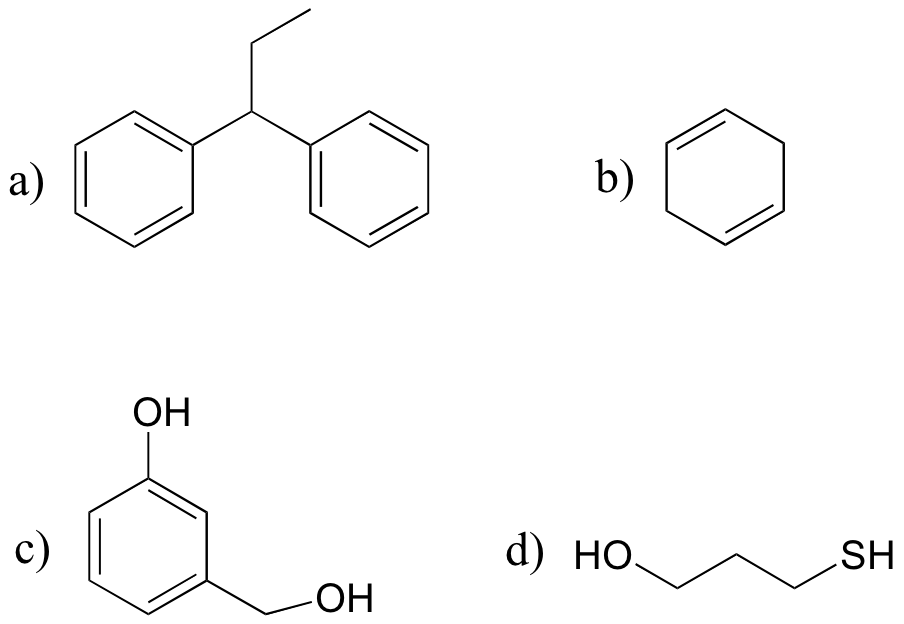
P17.4: Draw the radical intermediate species that you would expect to form when each of the compounds below reacts with a radical initiator.
P17.5: Azobis(isobutyronitrile) is a widely used radical initiator which rapidly undergoes homolytic decomposition when heated.
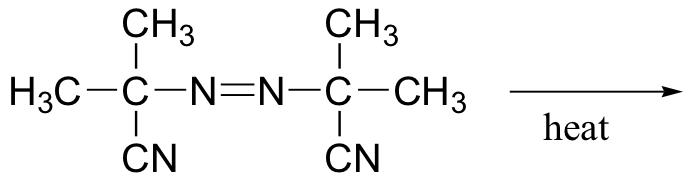
Predict the products of this decomposition reaction, and show a likely mechanism. What is the thermodynamic driving force for homolytic cleavage?
P17.6: A well-known transformation in the organic laboratory is the addition of chlorine or bromine to alkanes.
![]()
In the initiation phase of these reactions, Cl2 or Br2 undergoes homolytic cleavage in the presence of light or heat to give a halogen radical.
a) Draw a complete mechanism for the following reaction:
![]()
b) Under the same conditions, it is possible to get butane as an alternate product. Draw a mechanism showing how this occurs.
c) When 2-methylbutane is the alkyl starting material under the same conditions, a number of isomeric chloroalkanes with the formula C5H11Cl can form. Draw structures for these isomers, and for each draw the alkyl radical intermediate that led to its formation.
d) In part c), which is the most stable radical intermediate?
e) In the reaction in part c), is the most abundant chloroalkane necessarily the one that forms from the most stable radical intermediate? Explain.
P17.7: We learned in section 15.2B that HBr will react with alkenes in electrophilic addition reactions, and that these reactions have 'Markovnikov' regioselectivity. However, when the starting alkene contains even a small amount of contaminating peroxide (which happens when it is allowed to come into contact with air), a significant amount of 'anti-Markovnikov' product is often observed.
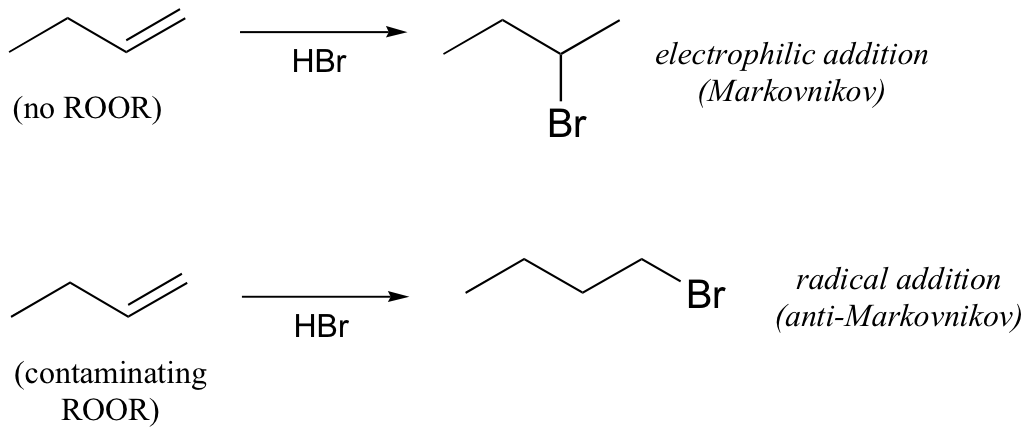
a) Propose a mechanism for formation of the anti-Markovnikov product.
b) Propose a mechanism for the anti-Markovnikov addition of thiol to an alkene (in the presence of peroxide)

P17.8: In section 12.4E we learned that aspirin works by blocking the action of an enzyme that catalyzes a key step in the biosynthesis of prostaglandins, a class of biochemical signaling molecules. The reaction in question is a dioxygenase, and involves several single-electron steps. First, arachidonate loses a single electron to an iron-bound oxygen radical in the enzyme. The arachidonate radical then reacts with molecular oxygen to form a five-membered oxygen-containing ring, followed by closure of a cyclopentane ring to yield yet another radical intermediate.
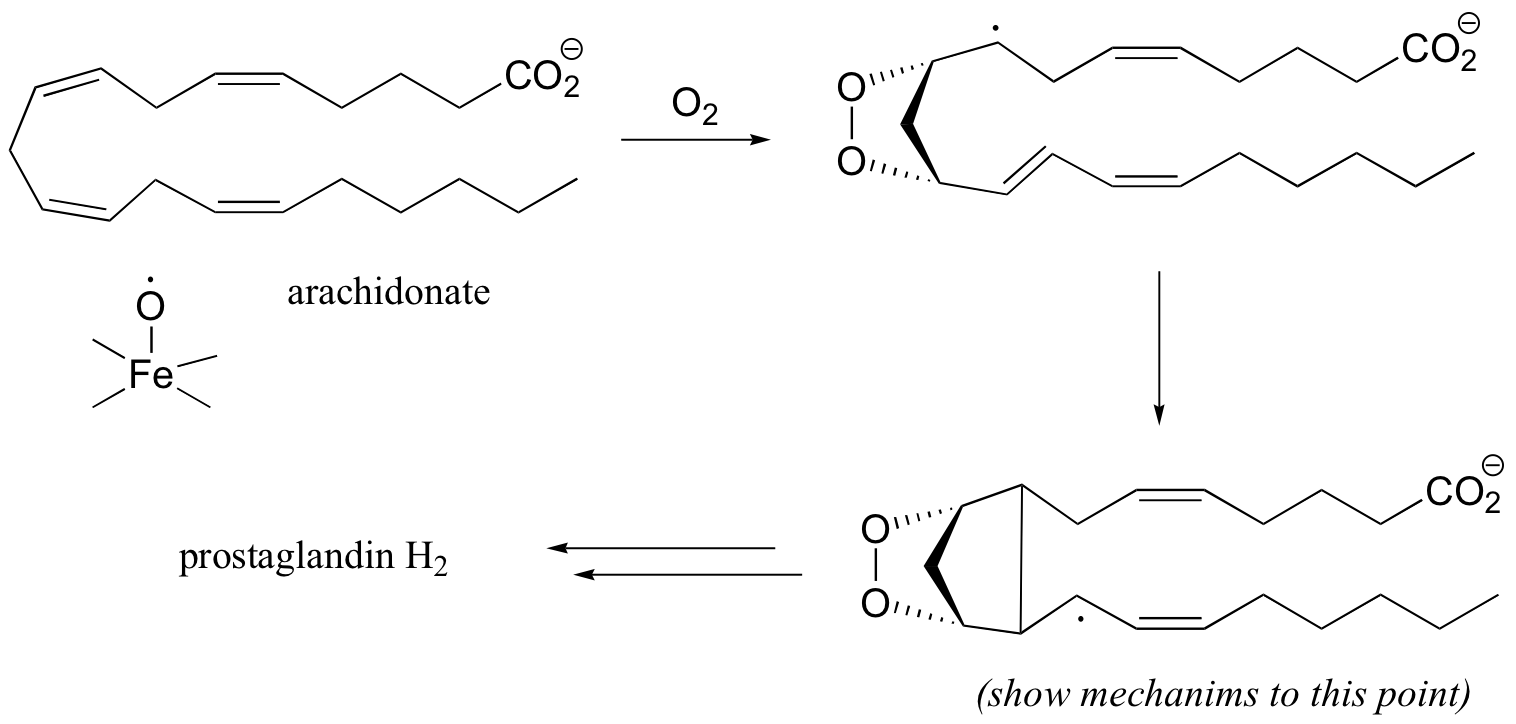
Propose a mechanism for the steps of the reaction that are shown in this figure.
P17.9: Anandamide, a derivative of arachidonate (see previous problem), is an important signaling molecule in humans.
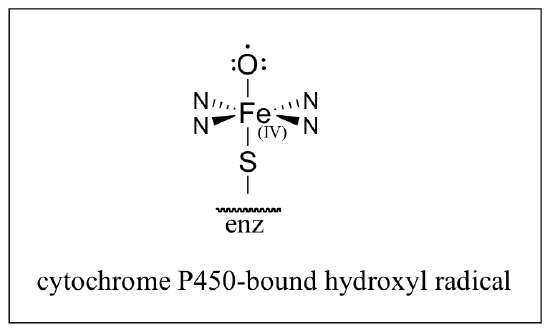
a) Recently it was shown that anandamide is a substrate for epoxidation by a cytochrome P450 monooxygenase (see section 17.3A). Starting with anandamide and the cytochrome P450-bound hydroxyl radical shown below, propose a (radical) mechanism for this reaction.
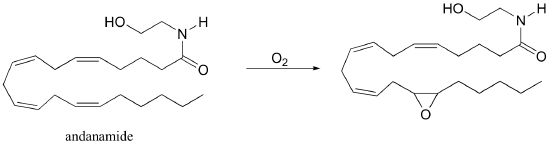
b) Anandamide also has shown to undergo cytochrome P450-dependant hydroxylation to form the product below.
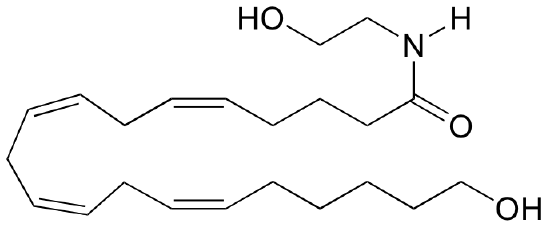
Propose a mechanism, starting with the same P450-bound hydroxyl-radical.
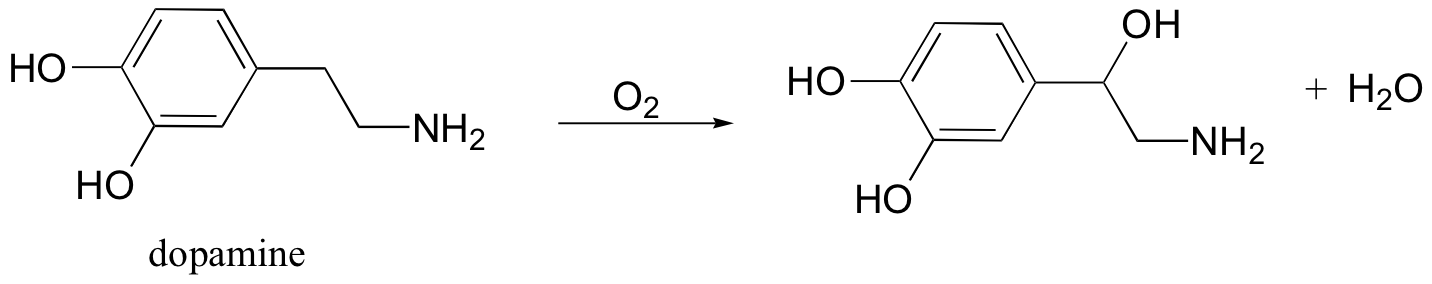
P17.10: Some redox enzymes use copper to assist in electron transfer steps. One important example is dopamine b-monooxygenase, which catalyzes the following reaction:
The following intermediates have been proposed (Biochemistry 1994, 33, 226):
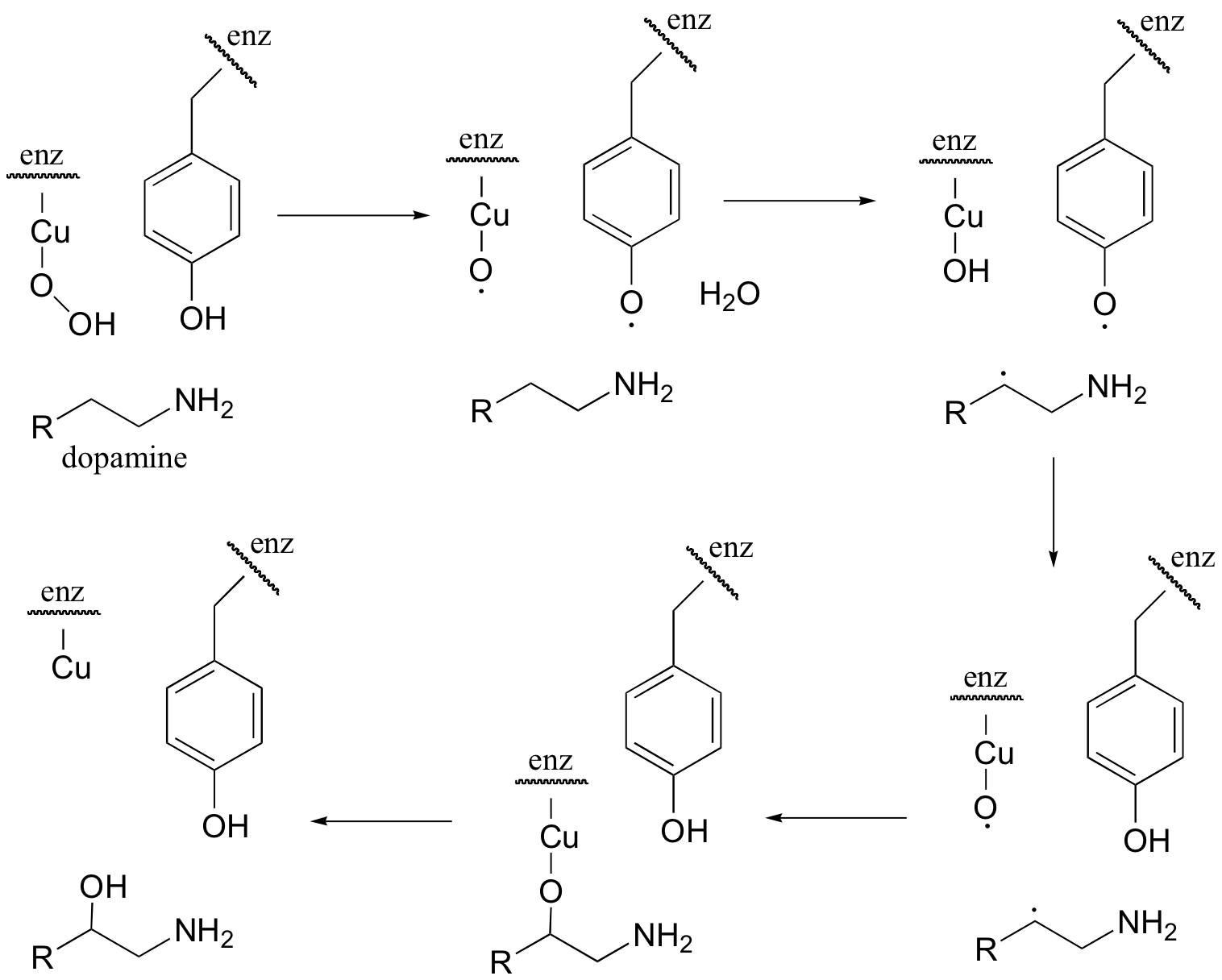
Draw mechanistic arrows to show how the reaction proceeds.
P17.11: Show a single-electron mechanism for the reaction FADH2 with (triplet) molecular oxygen to form flavin hydroperoxide. Assume that a semiquinone intermediate is involved.


