12.7: Grupos de amida activada
- Page ID
- 2406
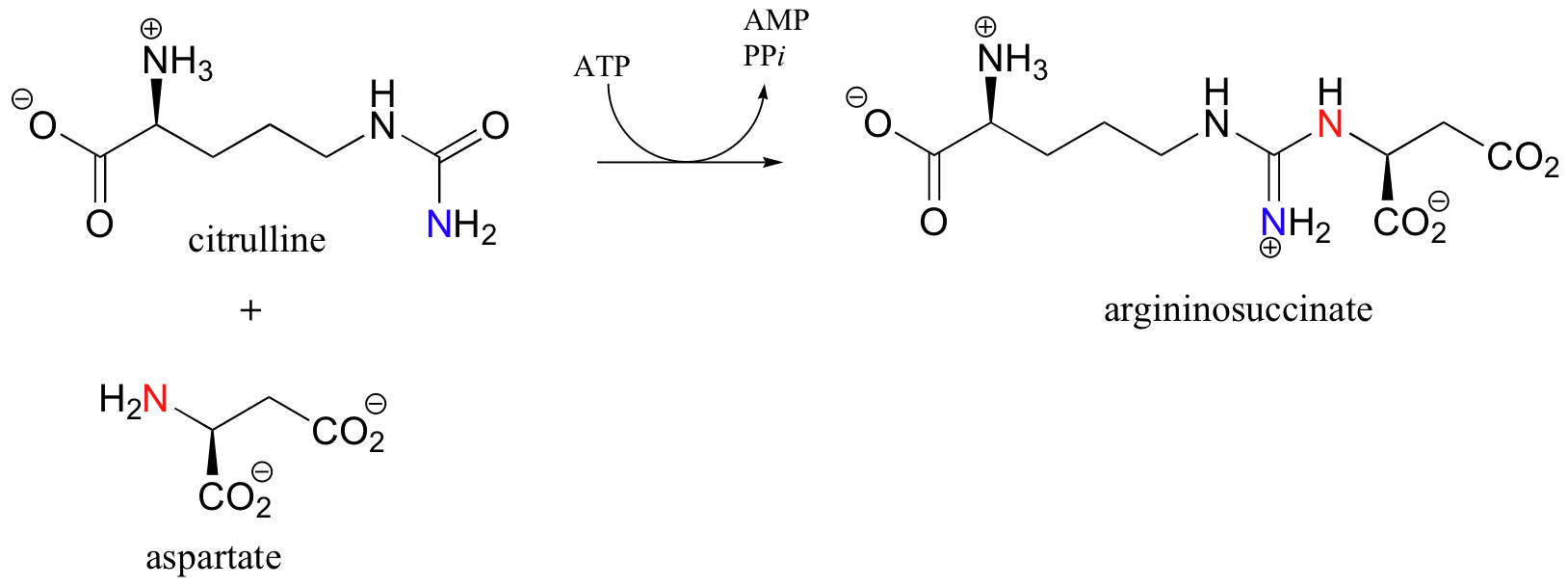
In discussing the nucleophilic acyl substitution reactions of acyl phosphates, thioester, esters, and amides, we have seen many slight variations on one overarching mechanistic theme. Let’s now look at a reaction that can be thought of as a ‘cousin’ of the nucleophilic acyl substitution, which follows the same general pattern but differs in several details. The reaction catalyzed by argininosuccinate synthetase is the second step in the urea cycle, a four-step series of reactions in which ammonia is converted into urea for elimination in urine.
3D Jmol model of argininosuccinate
Looking at this reaction, you can see that a substitution is occurring at an acyl group (an amide, to be specific), but that the atom that gets expelled is the amide oxygen rather than the nitrogen. As you know, amides are comparatively stable, and thus it stands to reason that the starting amide must be activated before the substitution reaction can take place. This occurs, as you might expect, through the formation of an acyl phosphate intermediate at the expense of one ATP molecule.
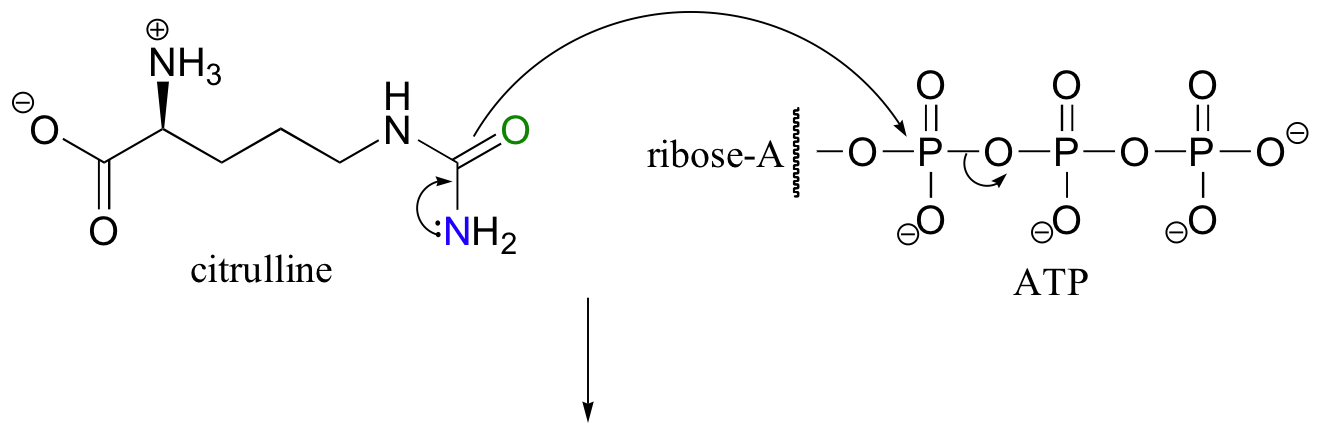
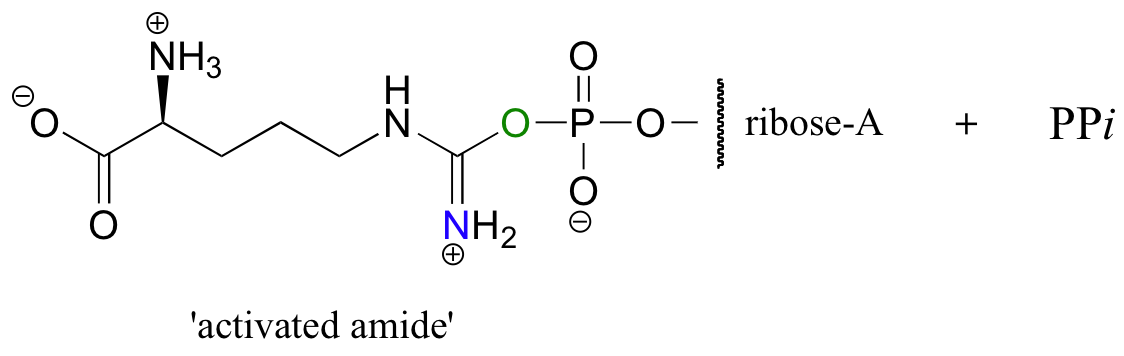
In this particular reaction, the amide oxygen acts as a nucleophile, attacking the alpha-phosphate of ATP to form the activated acyl-AMP intermediate. Next, a kind of acyl substitution occurs that we have not yet seen: the amine nitrogen on the aspartate attacks the electrophilic carbon of a carbon-nitrogen double bond, and the reaction proceeds through a tetrahedral intermediate before the AMP group is expelled, taking with it what was originally the carbonyl oxygen of the starting amide.


Look carefully at the progress of this reaction, and make sure that you can see how it fits the basic pattern of the other nucleophilic acyl substitution reactions we have studied.


